PMN-derived netrin-1 attenuates cardiac ischemia-reperfusion injury via myeloid ADORA2B signaling
- PMID: 33891683
- PMCID: PMC8077173
- DOI: 10.1084/jem.20210008
PMN-derived netrin-1 attenuates cardiac ischemia-reperfusion injury via myeloid ADORA2B signaling
Abstract
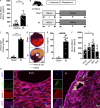
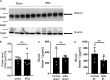
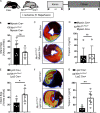
Previous studies implicated the neuronal guidance molecule netrin-1 in attenuating myocardial ischemia-reperfusion injury. However, the tissue-specific sources and receptor signaling events remain elusive. Neutrophils are among the first cells responding to an ischemic insult and can be associated with tissue injury or rescue. We found netrin-1 levels were elevated in the blood of patients with myocardial infarction, as well as in mice exposed to myocardial ischemia-reperfusion. Selectively increased infarct sizes and troponin levels were found in Ntn1loxP/loxP Lyz2 Cre+ mice, but not in mice with conditional netrin-1 deletion in other tissue compartments. In vivo studies using neutrophil depletion identified neutrophils as the main source for elevated blood netrin-1 during myocardial injury. Finally, pharmacologic studies using treatment with recombinant netrin-1 revealed a functional role for purinergic signaling events through the myeloid adenosine A2b receptor in mediating netrin-1-elicited cardioprotection. These findings suggest an autocrine signaling loop with a functional role for neutrophil-derived netrin-1 in attenuating myocardial ischemia-reperfusion injury through myeloid adenosine A2b signaling.
© 2021 Li et al.
Conflict of interest statement
Disclosures: The authors declare no competing interests exist.
Figures








Comment in
-
Immune cells-A curse and a blessing!J Exp Med. 2021 Jun 7;218(6):e20210590. doi: 10.1084/jem.20210590. Epub 2021 May 21. J Exp Med. 2021. PMID: 34019638 Free PMC article.
References
-
- Aherne, C.M., Collins C.B., Masterson J.C., Tizzano M., Boyle T.A., Westrich J.A., Parnes J.A., Furuta G.T., Rivera-Nieves J., and Eltzschig H.K.. 2012. Neuronal guidance molecule netrin-1 attenuates inflammatory cell trafficking during acute experimental colitis. Gut. 61:695–705. 10.1136/gutjnl-2011-300012 - DOI - PMC - PubMed
-
- Aherne, C.M., Saeedi B., Collins C.B., Masterson J.C., McNamee E.N., Perrenoud L., Rapp C.R., Curtis V.F., Bayless A., Fletcher A., et al. . 2015. Epithelial-specific A2B adenosine receptor signaling protects the colonic epithelial barrier during acute colitis. Mucosal Immunol. 8:1324–1338. 10.1038/mi.2015.22 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

