Intermittent hypoxia-induced downregulation of microRNA-320b promotes lung cancer tumorigenesis by increasing CDT1 via USP37
- PMID: 33898105
- PMCID: PMC8056179
- DOI: 10.1016/j.omtn.2020.12.023
Intermittent hypoxia-induced downregulation of microRNA-320b promotes lung cancer tumorigenesis by increasing CDT1 via USP37
Abstract
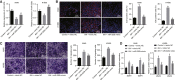
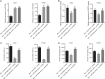
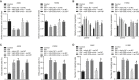
Obstructive sleep apnea-hypopnea (OSAH) is correlated with an increased incidence of lung cancer. In our study, we explored the functional roles of microRNAs (miRNAs) in lung cancer patients that were complicated with OSAH involving the deubiquitination enzyme. The miR-320b expression pattern in lung cancer tissues and cells was determined. The interactions between ubiquitin-specific peptidase 37 (USP37) and miR-320b were evaluated by a dual-luciferase reporter gene assay, whereas USP37 and Cdc10-dependent transcript 1 (CDT1) was assessed by co-immunoprecipitation and immunofluorescence. After the induction of intermittent hypoxia (IH), a gain-of function approach was performed to investigate roles of miR-320b, USP37, and CDT1 in lung cancer cell proliferation and invasion. In addition, nude mouse xenograft models were used to study their effects on tumor growth in vivo. miR-320b was poorly expressed in lung cancer patients with OSAH. IH treatment downregulated the expression of miR-320b but promoted the proliferation and invasion capabilities of lung cancer cells, both of which were suppressed by the overexpression of miR-320b through decreasing USP37. USP37 interacted with and deubiquitinated CDT1 to protect it from proteasomal degradation. Our study uncovered that IH-induced downregulation of miR-320b promoted the tumorigenesis of lung cancer by the USP37-mediated deubiquitination of CDT1.
Keywords: CDT1; USP37; deubiquitinase; lung cancer; microRNA-320b; obstructive sleep apnea-hypopnea.
© 2021 The Authors.
Conflict of interest statement
The authors declare no competing interests.
Figures






Similar articles
-
microRNA-320b suppresses HNF4G and IGF2BP2 expression to inhibit angiogenesis and tumor growth of lung cancer.Carcinogenesis. 2021 May 28;42(5):762-771. doi: 10.1093/carcin/bgab023. Carcinogenesis. 2021. PMID: 33758932
-
USP37 deubiquitinates Cdt1 and contributes to regulate DNA replication.Mol Oncol. 2016 Oct;10(8):1196-206. doi: 10.1016/j.molonc.2016.05.008. Epub 2016 Jun 3. Mol Oncol. 2016. PMID: 27296872 Free PMC article.
-
miR-320b suppresses cell proliferation by targeting c-Myc in human colorectal cancer cells.BMC Cancer. 2015 Oct 20;15:748. doi: 10.1186/s12885-015-1728-5. BMC Cancer. 2015. PMID: 26487644 Free PMC article.
-
Long Noncoding RNA XIST Promotes Osteosarcoma Progression by Targeting Ras-Related Protein RAP2B via miR-320b.Oncol Res. 2018 Jul 5;26(6):837-846. doi: 10.3727/096504017X14920318811721. Epub 2017 Apr 12. Oncol Res. 2018. PMID: 28409547 Free PMC article.
-
Ubiquitin-specific peptidase 37: an important cog in the oncogenic machinery of cancerous cells.J Exp Clin Cancer Res. 2021 Nov 10;40(1):356. doi: 10.1186/s13046-021-02163-7. J Exp Clin Cancer Res. 2021. PMID: 34758854 Free PMC article. Review.
Cited by
-
Ubiquitin specific peptidase 37 and PCNA interaction promotes osteosarcoma pathogenesis by modulating replication fork progression.J Transl Med. 2023 Apr 28;21(1):286. doi: 10.1186/s12967-023-04126-2. J Transl Med. 2023. PMID: 37118828 Free PMC article.
-
Identification of hypoxia-related gene signatures based on multi-omics analysis in lung adenocarcinoma.J Cell Mol Med. 2024 Jan;28(2):e18032. doi: 10.1111/jcmm.18032. Epub 2023 Nov 27. J Cell Mol Med. 2024. PMID: 38013642 Free PMC article.
-
Identification of CDT1 as a prognostic marker in human lung adenocarcinoma using bioinformatics approaches.PeerJ. 2023 Sep 28;11:e16166. doi: 10.7717/peerj.16166. eCollection 2023. PeerJ. 2023. PMID: 37790630 Free PMC article.
-
Chromatin licensing and DNA replication factor 1 as a potential prognostic and diagnostic biomarker for gastric cancer.Sci Rep. 2024 Dec 28;14(1):31000. doi: 10.1038/s41598-024-81977-9. Sci Rep. 2024. PMID: 39730687 Free PMC article.
-
CDK1-mediated phosphorylation of USP37 regulates SND1 stability and promotes oncogenesis in colorectal cancer.Acta Pharm Sin B. 2025 Apr;15(4):1938-1955. doi: 10.1016/j.apsb.2025.02.014. Epub 2025 Feb 18. Acta Pharm Sin B. 2025. PMID: 40486858 Free PMC article.
References
-
- Hirsch F.R., Scagliotti G.V., Mulshine J.L., Kwon R., Curran W.J., Jr., Wu Y.L., Paz-Ares L. Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389:299–311. - PubMed
-
- Ren W., Mi D., Yang K., Cao N., Tian J., Li Z., Ma B. The expression of hypoxia-inducible factor-1α and its clinical significance in lung cancer: a systematic review and meta-analysis. Swiss Med. Wkly. 2013;143:w13855. - PubMed
-
- Wilson W.R., Hay M.P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer. 2011;11:393–410. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

