GZ17-6.02 Interacts With [MEK1/2 and B-RAF Inhibitors] to Kill Melanoma Cells
- PMID: 33898322
- PMCID: PMC8061416
- DOI: 10.3389/fonc.2021.656453
GZ17-6.02 Interacts With [MEK1/2 and B-RAF Inhibitors] to Kill Melanoma Cells
Abstract
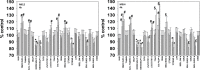
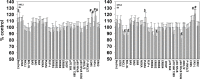
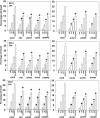
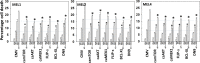
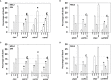
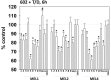
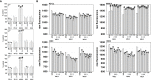
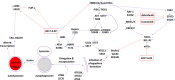
We defined the lethal interaction between the novel therapeutic GZ17-6.02 and the standard of care combination of the MEK1/2 inhibitor trametinib and the B-RAF inhibitor dabrafenib in PDX isolates of cutaneous melanoma expressing a mutant B-RAF V600E protein. GZ17-6.02 interacted with trametinib/dabrafenib in an additive fashion to kill melanoma cells. Regardless of prior vemurafenib resistance, the drugs when combined interacted to prolong ATM S1981/AMPK T172 and eIF2α S51 phosphorylation and prolong the reduced phosphorylation of JAK2 Y1007, STAT3 Y705 and STAT5 Y694. In vemurafenib-resistant cells GZ17-6.02 caused a prolonged reduction in mTORC1 S2448, mTORC2 S2481 and ULK1 S757 phosphorylation; regardless of vemurafenib resistance, GZ17-6.02 caused a prolonged elevation in CD95 and FAS-L expression. Knock down of eIF2α, Beclin1, ATG5, ATM, AMPKα, CD95 or FADD significantly reduced the ability of GZ17-6.02 to kill as a single agent or when combined with the kinase inhibitors. Expression of activated mTOR, activated STAT3, activated MEK1 or activated AKT significantly reduced the ability of GZ17-6.02 to kill as a single agent or when combined with kinase inhibitors; protective effects that were significantly less pronounced in cells treated with trametinib/dabrafenib. Regardless of vemurafenib resistance, the drugs alone or in combination all reduced the expression of PD-L1 and increased the levels of MHCA, which was linked to degradation of multiple HDAC proteins. Our findings support the use of GZ17-6.02 in combination with trametinib/dabrafenib in the treatment of melanomas expressing mutant B-RAF V600E proteins.
Keywords: B-Raf mutation V600E; GZ17-6.02; HDAC; autophagy; er stress.
Copyright © 2021 Booth, West, Von Hoff, Kirkwood and Dent.
Conflict of interest statement
PD has received funding support from Genzada Pharmaceuticals Inc. for these studies. CW is a paid officer of the company. PD and DV are Consultants and Key Scientific advisors to the company. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

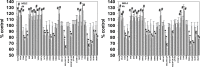

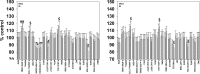
Similar articles
-
GZ17-6.02 and axitinib interact to kill renal carcinoma cells.Oncotarget. 2022 Feb 4;13:281-290. doi: 10.18632/oncotarget.28189. eCollection 2022. Oncotarget. 2022. PMID: 35136485 Free PMC article.
-
GZ17-6.02 and Pemetrexed Interact to Kill Osimertinib-Resistant NSCLC Cells That Express Mutant ERBB1 Proteins.Front Oncol. 2021 Aug 19;11:711043. doi: 10.3389/fonc.2021.711043. eCollection 2021. Front Oncol. 2021. PMID: 34490108 Free PMC article.
-
GZ17-6.02 kills PDX isolates of uveal melanoma.Oncotarget. 2024 May 17;15:328-344. doi: 10.18632/oncotarget.28586. Oncotarget. 2024. PMID: 38758815 Free PMC article.
-
Targeting oncogenic Raf protein-serine/threonine kinases in human cancers.Pharmacol Res. 2018 Sep;135:239-258. doi: 10.1016/j.phrs.2018.08.013. Epub 2018 Aug 15. Pharmacol Res. 2018. PMID: 30118796 Review.
-
Allosteric MEK1/2 inhibitors including cobimetanib and trametinib in the treatment of cutaneous melanomas.Pharmacol Res. 2017 Mar;117:20-31. doi: 10.1016/j.phrs.2016.12.009. Epub 2016 Dec 9. Pharmacol Res. 2017. PMID: 27956260 Review.
Cited by
-
GZ17-6.02 interacts with bexarotene to kill mycosis fungoides cells.Oncotarget. 2024 Feb 8;15:124-133. doi: 10.18632/oncotarget.28557. Oncotarget. 2024. PMID: 38329728 Free PMC article.
-
GZ17-6.02 and palbociclib interact to kill ER+ breast cancer cells.Oncotarget. 2022 Jan 11;13:92-104. doi: 10.18632/oncotarget.28177. eCollection 2022. Oncotarget. 2022. PMID: 35035775 Free PMC article.
-
Histone deacetylases in the regulation of cell death and survival mechanisms in resistant BRAF-mutant cancers.Cancer Drug Resist. 2025 Jan 25;8:6. doi: 10.20517/cdr.2024.125. eCollection 2025. Cancer Drug Resist. 2025. PMID: 39935431 Free PMC article. Review.
-
GZ17-6.02 Inhibits the Growth of EGFRvIII+ Glioblastoma.Int J Mol Sci. 2022 Apr 10;23(8):4174. doi: 10.3390/ijms23084174. Int J Mol Sci. 2022. PMID: 35456993 Free PMC article.
-
Transcriptomic and proteomic analysis of tumor suppressive effects of GZ17-6.02 against mycosis fungoides.Sci Rep. 2024 Jan 23;14(1):1955. doi: 10.1038/s41598-024-52544-z. Sci Rep. 2024. PMID: 38263212 Free PMC article.
References
-
- Moser JC, Chen D, Hu-Lieskovan S, Grossmann KF, Patel S, Colonna SV, et al. . Real-world survival of patients with advanced BRAF V600 mutated melanoma treated with front-line BRAF/MEK inhibitors, anti-PD-1 antibodies, or nivolumab/ipilimumab. Cancer Med (2019) 8:7637–43. 10.1002/cam4.2625 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

