Discovery of Kasugamycin as a Potent Inhibitor of Glycoside Hydrolase Family 18 Chitinases
- PMID: 33898519
- PMCID: PMC8058351
- DOI: 10.3389/fmolb.2021.640356
Discovery of Kasugamycin as a Potent Inhibitor of Glycoside Hydrolase Family 18 Chitinases
Abstract
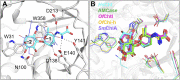
Kasugamycin, a well-known aminoglycoside antibiotic, has been used widely in agriculture and medicine to combat microbial pathogens by binding the ribosome to inhibit translation. Here, kasugamycin was discovered to be a competitive inhibitor of glycoside hydrolase family 18 (GH18) chitinases from three different organisms (bacterium, insect and human). Results from tryptophan fluorescence spectroscopy and molecular docking revealed that kasugamycin binds to the substrate-binding clefts in a similar mode as the substrate. An electrostatic interaction between the amino group of kasugamycin and the carboxyl group of a conserved aspartate in GH18 chitinase (one of the catalytic triad residues) was found to be vital for the inhibitory activity. This paper not only reports new molecular targets of kasugamycin, but also expands our thinking about GH inhibitor design by using a scaffold unrelated to the substrate.
Keywords: chitinase; glycoside hydrolase; inhibitor; kasugamycin; target.
Copyright © 2021 Qi, Jiang, Ding, Liu and Yang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
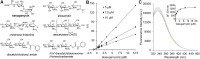
Similar articles
-
Glycoside hydrolase family 18 and 20 enzymes are novel targets of the traditional medicine berberine.J Biol Chem. 2018 Oct 5;293(40):15429-15438. doi: 10.1074/jbc.RA118.004351. Epub 2018 Aug 22. J Biol Chem. 2018. PMID: 30135205 Free PMC article.
-
Glycoside hydrolase family 18 chitinases: The known and the unknown.Biotechnol Adv. 2020 Nov 1;43:107553. doi: 10.1016/j.biotechadv.2020.107553. Epub 2020 May 19. Biotechnol Adv. 2020. PMID: 32439576 Review.
-
Crystal structure-guided design of berberine-based novel chitinase inhibitors.J Enzyme Inhib Med Chem. 2020 Dec;35(1):1937-1943. doi: 10.1080/14756366.2020.1837123. J Enzyme Inhib Med Chem. 2020. PMID: 33167737 Free PMC article.
-
Versatile glycoside hydrolase family 18 chitinases for fungi ingestion and reproduction in the pinewood nematode Bursaphelenchus xylophilus.Int J Parasitol. 2016 Nov;46(12):819-828. doi: 10.1016/j.ijpara.2016.08.001. Epub 2016 Sep 15. Int J Parasitol. 2016. PMID: 27641827
-
Chitinolytic enzymes: catalysis, substrate binding, and their application.Curr Protein Pept Sci. 2000 Jul;1(1):105-24. doi: 10.2174/1389203003381450. Curr Protein Pept Sci. 2000. PMID: 12369923 Review.
Cited by
-
Host chitinase 3-like-1 is a universal therapeutic target for SARS-CoV-2 viral variants in COVID-19.Elife. 2022 Jun 23;11:e78273. doi: 10.7554/eLife.78273. Elife. 2022. PMID: 35735790 Free PMC article.
-
Effects of chitinase-1 inhibitor in obesity-induced and -aggravated asthma in a murine model.Life Sci. 2023 Dec 1;334:122163. doi: 10.1016/j.lfs.2023.122163. Epub 2023 Oct 25. Life Sci. 2023. PMID: 37890698 Free PMC article.
-
Discovery of conformation constrained tetracyclic compounds as potent chitinase OfChi-h inhibitors with a novel binding mode.J Enzyme Inhib Med Chem. 2025 Dec;40(1):2528056. doi: 10.1080/14756366.2025.2528056. Epub 2025 Jul 7. J Enzyme Inhib Med Chem. 2025. PMID: 40619956 Free PMC article.
-
Streptomyces and their specialised metabolites for phytopathogen control - comparative in vitro and in planta metabolic approaches.Front Plant Sci. 2023 Jun 14;14:1151912. doi: 10.3389/fpls.2023.1151912. eCollection 2023. Front Plant Sci. 2023. PMID: 37389291 Free PMC article. Review.
-
Microbial production of 5-epi-jinkoheremol, a plant-derived antifungal sesquiterpene.Appl Environ Microbiol. 2024 Oct 23;90(10):e0119124. doi: 10.1128/aem.01191-24. Epub 2024 Sep 16. Appl Environ Microbiol. 2024. PMID: 39283105 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

