CD19 CAR-T cell treatment conferred sustained remission in B-ALL patients with minimal residual disease
- PMID: 33899130
- PMCID: PMC8571234
- DOI: 10.1007/s00262-021-02941-4
CD19 CAR-T cell treatment conferred sustained remission in B-ALL patients with minimal residual disease
Abstract
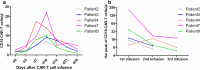
The persistence or recurrence of minimal residual disease (MRD) after chemotherapy predicts relapse of B-cell acute lymphoblastic leukemia (B-ALL). CD19-directed chimeric antigen receptor T (CD19 CAR-T) cells have shown promising responses in B-ALL. However, their role in chemotherapy-refractory MRD-positive B-ALL remains unclear. Here we aimed to assess the effectiveness and safety of CD19 CAR-T cells in MRD-positive B-ALL patients. From January 2018, a total of 14 MRD-positive B-ALL patients received one or more infusions of autogenous CD19 CAR-T cells. Among them, 12 patients achieved MRD-negative remission after one cycle of CAR-T infusion. At a median follow-up time of 647 days (range 172-945 days), the 2-year event-free survival rate in MRD-positive patients was 61.2% ± 14.0% and the 2-year overall survival was 78.6 ± 11.0%, which were significantly higher than patients with active disease (blasts ≥ 5% or with extramedullary disease). Moreover, patients with MRD had a lower grade of cytokine release syndrome (CRS) than patients with active disease. However, the peak expansion of CAR-T cells in MRD positive patients showed no statistical difference compared to patients with active disease. Five patients received two or more CAR-T cell infusions and these patients showed a decreased peak expansion of CAR-T cell in subsequent infusions. In conclusion, pre-emptive CD19 CAR-T cell treatment is an effective and safe approach and may confer sustained remission in B-ALL patients with chemotherapy-refractory MRD. The trials were registered at www.chictr.org.cn as ChiCTR-ONN-16009862 (November 14, 2016) and ChiCTR1800015164 (March 11, 2018).
Keywords: B-cell acute lymphoblastic leukemia; CD19-directed chimeric antigen receptor T cells; Cytokine release syndrome; Minimal residual disease; Relapse.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interests.
Figures


Similar articles
-
CD19-Specific CAR-T Cell Treatment of 115 Children and Young Adults with Acute B Lymphoblastic Leukemia: Long-term Follow-up.Cancer Res Treat. 2024 Jul;56(3):945-955. doi: 10.4143/crt.2023.1205. Epub 2024 Feb 13. Cancer Res Treat. 2024. PMID: 38351683 Free PMC article.
-
Low Peripheral Blood Counts and Elevated Proinflammatory Cytokines Signal a Poor CD19 Chimeric Antigen Receptor T-cell Response in Acute Lymphoblastic Leukemia.Transplant Cell Ther. 2025 Aug;31(8):551-564. doi: 10.1016/j.jtct.2025.05.003. Epub 2025 May 20. Transplant Cell Ther. 2025. PMID: 40398620
-
Evidence of long-lasting anti-CD19 activity of engrafted CD19 chimeric antigen receptor-modified T cells in a phase I study targeting pediatrics with acute lymphoblastic leukemia.Hematol Oncol. 2019 Dec;37(5):601-608. doi: 10.1002/hon.2672. Epub 2019 Sep 15. Hematol Oncol. 2019. PMID: 31465532 Free PMC article. Clinical Trial.
-
Systematic Review and Meta-analysis of CD19-Specific CAR-T Cell Therapy in Relapsed/Refractory Acute Lymphoblastic Leukemia in the Pediatric and Young Adult Population: Safety and Efficacy Outcomes.Clin Lymphoma Myeloma Leuk. 2021 Apr;21(4):e334-e347. doi: 10.1016/j.clml.2020.12.010. Epub 2020 Dec 17. Clin Lymphoma Myeloma Leuk. 2021. PMID: 33573914
-
Chimeric Antigen Receptor T Cell Therapy for Pediatric B-ALL: Narrowing the Gap Between Early and Long-Term Outcomes.Front Immunol. 2020 Aug 11;11:1985. doi: 10.3389/fimmu.2020.01985. eCollection 2020. Front Immunol. 2020. PMID: 32849662 Free PMC article. Review.
Cited by
-
Prophylactic donor-derived CD19 CAR-T cell infusion for preventing relapse in high-risk B-ALL after allogeneic hematopoietic stem cell transplantation.Leukemia. 2024 Jun;38(6):1419-1422. doi: 10.1038/s41375-024-02251-5. Epub 2024 Apr 17. Leukemia. 2024. PMID: 38632315 Free PMC article. No abstract available.
-
Early T-Cell Precursor Acute Lymphoblastic Leukemia: Diagnosis, Updates in Molecular Pathogenesis, Management, and Novel Therapies.Front Oncol. 2021 Nov 29;11:750789. doi: 10.3389/fonc.2021.750789. eCollection 2021. Front Oncol. 2021. PMID: 34912707 Free PMC article. Review.
-
Hyper-CVAD-Based Stem Cell Microtransplant as Post-Remission Therapy in Acute Lymphoblastic Leukemia.Stem Cells Transl Med. 2022 Nov 18;11(11):1113-1122. doi: 10.1093/stcltm/szac066. Stem Cells Transl Med. 2022. PMID: 36181762 Free PMC article.
-
Challenges and Advances in Chimeric Antigen Receptor Therapy for Acute Myeloid Leukemia.Cancers (Basel). 2022 Jan 19;14(3):497. doi: 10.3390/cancers14030497. Cancers (Basel). 2022. PMID: 35158765 Free PMC article. Review.
-
Role of allogeneic haematopoietic stem cell transplantation in the treatment of adult acute lymphoblastic leukaemia in the era of immunotherapy.Chin Med J (Engl). 2022 Apr 20;135(8):890-900. doi: 10.1097/CM9.0000000000001898. Chin Med J (Engl). 2022. PMID: 34890382 Free PMC article. Review.
References
-
- Bassan R, Bruggemann M, Radcliffe HS, Hartfield E, Kreuzbauer G, Wetten S. A systematic literature review and meta-analysis of minimal residual disease as a prognostic indicator in adult B-cell acute lymphoblastic leukemia. Haematologica. 2019;104(10):2028–2039. doi: 10.3324/haematol.2018.201053. - DOI - PMC - PubMed
-
- Raff T, Gokbuget N, Luschen S, Reutzel R, Ritgen M, Irmer S, Bottcher S, Horst HA, Kneba M, Hoelzer D, Bruggemann M, Group GS Molecular relapse in adult standard-risk ALL patients detected by prospective MRD monitoring during and after maintenance treatment: data from the GMALL 06/99 and 07/03 trials. Blood. 2007;109(3):910–915. doi: 10.1182/blood-2006-07-037093. - DOI - PubMed
-
- He X, Xiao X, Li Q, Jiang Y, Cao Y, Sun R, Jin X, Yuan T, Meng J, Ma L, Lu W, Lyu C, Liu K, Zhao M. Anti-CD19 CAR-T as a feasible and safe treatment against central nervous system leukemia after intrathecal chemotherapy in adults with relapsed or refractory B-ALL [letter] Leukemia. 2019;33(8):2102–2104. doi: 10.1038/s41375-019-0437-5. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

