Exposing unsaturated Cu1-O2 sites in nanoscale Cu-MOF for efficient electrocatalytic hydrogen evolution
- PMID: 33910899
- PMCID: PMC8081372
- DOI: 10.1126/sciadv.abg2580
Exposing unsaturated Cu1-O2 sites in nanoscale Cu-MOF for efficient electrocatalytic hydrogen evolution
Abstract
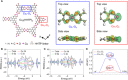
Conductive metal-organic framework (MOF) materials have been recently considered as effective electrocatalysts. However, they usually suffer from two major drawbacks, poor electrochemical stability and low electrocatalytic activity in bulk form. Here, we have developed a rational strategy to fabricate a promising electrocatalyst composed of a nanoscale conductive copper-based MOF (Cu-MOF) layer fully supported over synergetic iron hydr(oxy)oxide [Fe(OH) x ] nanoboxes. Owing to the highly exposed active centers, enhanced charge transfer, and robust hollow nanostructure, the obtained Fe(OH) x @Cu-MOF nanoboxes exhibit superior activity and stability for the electrocatalytic hydrogen evolution reaction (HER). Specifically, it needs an overpotential of 112 mV to reach a current density of 10 mA cm-2 with a small Tafel slope of 76 mV dec-1 X-ray absorption fine structure spectroscopy combined with density functional theory calculations unravels that the highly exposed coordinatively unsaturated Cu1-O2 centers could effectively accelerate the formation of key *H intermediates toward fast HER kinetics.
Copyright © 2021 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
Figures




References
-
- Zimmerman J. B., Anastas P. T., Erythropel H. C., Leitner W., Designing for a green chemistry future. Science 367, 397–400 (2020). - PubMed
-
- Zhang H., Yu L., Chen T., Zhou W., Lou X. W., Surface modulation of hierarchical MoS2 nanosheets by Ni single atoms for enhanced electrocatalytic hydrogen evolution. Adv. Funct. Mater. 28, 1807086 (2018).
-
- Zhu J., Hu L., Zhao P., Lee L. Y. S., Wong K.-Y., Recent advances in electrocatalytic hydrogen evolution using nanoparticles. Chem. Rev. 120, 851–918 (2020). - PubMed
-
- Zhang H., Nai J., Yu L., Lou X. W., Metal-organic-framework-based materials as platforms for renewable energy and environmental applications. Joule 1, 77–107 (2017).
-
- Lu X. F., Yu L., Zhang J., Lou X. W., Ultrafine dual-phased carbide nanocrystals confined in porous nitrogen-doped carbon dodecahedrons for efficient hydrogen evolution reaction. Adv. Mater. 31, 1900699 (2019). - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

