Altered Relationship Between Parvalbumin and Perineuronal Nets in an Autism Model
- PMID: 33912009
- PMCID: PMC8072465
- DOI: 10.3389/fnmol.2021.597812
Altered Relationship Between Parvalbumin and Perineuronal Nets in an Autism Model
Abstract
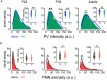
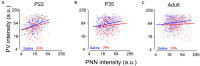
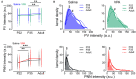
Altered function or presence of inhibitory neurons is documented in autism spectrum disorders (ASD), but the mechanism underlying this alternation is poorly understood. One major subtype of inhibitory neurons altered is the parvalbumin (PV)-containing neurons with reduced density and intensity in ASD patients and model mice. A subpopulation of PV+ neurons expresses perineuronal nets (PNN). To better understand whether the relationship between PV and PNN is altered in ASD, we measured quantitatively the intensities of PV and PNN in single PV+ neurons in the prelimbic prefrontal cortex (PrL-PFC) of a valproic acid (VPA) model of ASD at different ages. We found a decreased PV intensity but increased PNN intensity in VPA mice. The relationship between PV and PNN intensities is altered in VPA mice, likely due to an "abnormal" subpopulation of neurons with an altered PV-PNN relationship. Furthermore, reducing PNN level using in vivo injection of chondroitinase ABC corrects the PV expression in adult VPA mice. We suggest that the interaction between PV and PNN is disrupted in PV+ neurons in VPA mice which may contribute to the pathology in ASD.
Keywords: autism spectrum disorder; chondroitinase ABC; parvalbumin; perineuronal net; valproic acid.
Copyright © 2021 Xia, Li, Yang and Zhou.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Impact of Perineuronal Net Removal in the Rat Medial Prefrontal Cortex on Parvalbumin Interneurons After Reinstatement of Cocaine Conditioned Place Preference.Front Cell Neurosci. 2022 Jul 28;16:932391. doi: 10.3389/fncel.2022.932391. eCollection 2022. Front Cell Neurosci. 2022. PMID: 35966203 Free PMC article.
-
Post-weaning social isolation in male mice leads to abnormal aggression and disrupted network organization in the prefrontal cortex: Contribution of parvalbumin interneurons with or without perineuronal nets.Neurobiol Stress. 2023 May 29;25:100546. doi: 10.1016/j.ynstr.2023.100546. eCollection 2023 Jul. Neurobiol Stress. 2023. PMID: 37323648 Free PMC article.
-
Prenatal Valproate Exposure Differentially Affects Parvalbumin-Expressing Neurons and Related Circuits in the Cortex and Striatum of Mice.Front Mol Neurosci. 2016 Dec 21;9:150. doi: 10.3389/fnmol.2016.00150. eCollection 2016. Front Mol Neurosci. 2016. PMID: 28066177 Free PMC article.
-
The Perineuronal 'Safety' Net? Perineuronal Net Abnormalities in Neurological Disorders.Front Mol Neurosci. 2018 Aug 3;11:270. doi: 10.3389/fnmol.2018.00270. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30123106 Free PMC article. Review.
-
Proteolytic Remodeling of Perineuronal Nets: Effects on Synaptic Plasticity and Neuronal Population Dynamics.Neural Plast. 2018 Feb 4;2018:5735789. doi: 10.1155/2018/5735789. eCollection 2018. Neural Plast. 2018. PMID: 29531525 Free PMC article. Review.
Cited by
-
Novel ANKRD17 variants implicate synaptic and mitochondrial disruptions in intellectual disability and autism spectrum disorder.J Neurodev Disord. 2025 Jul 2;17(1):36. doi: 10.1186/s11689-025-09619-3. J Neurodev Disord. 2025. PMID: 40604385 Free PMC article.
-
The Pathophysiological Underpinnings of Gamma-Band Alterations in Psychiatric Disorders.Life (Basel). 2024 Apr 30;14(5):578. doi: 10.3390/life14050578. Life (Basel). 2024. PMID: 38792599 Free PMC article. Review.
-
Long-Term Enhancement of NMDA Receptor Function in Inhibitory Neurons Preferentially Modulates Potassium Channels and Cell Adhesion Molecules.Front Pharmacol. 2022 Jan 4;12:796179. doi: 10.3389/fphar.2021.796179. eCollection 2021. Front Pharmacol. 2022. PMID: 35058780 Free PMC article.
-
Region-Specific Alterations of Perineuronal Net Expression in Postmortem Autism Brain Tissue.Front Mol Neurosci. 2022 Apr 13;15:838918. doi: 10.3389/fnmol.2022.838918. eCollection 2022. Front Mol Neurosci. 2022. PMID: 35493330 Free PMC article.
-
Brevican, Neurocan, Tenascin-C, and Tenascin-R Act as Important Regulators of the Interplay Between Perineuronal Nets, Synaptic Integrity, Inhibitory Interneurons, and Otx2.Front Cell Dev Biol. 2022 Jun 2;10:886527. doi: 10.3389/fcell.2022.886527. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35721494 Free PMC article.
References
-
- Bringas M. E., Carvajal-Flores F. N., López-Ramírez T. A., Atzori M., Flores G. (2013). Rearrangement of the dendritic morphology in limbic regions and altered exploratory behavior in a rat model of autism spectrum disorder. Neuroscience 241, 170–187. 10.1016/j.neuroscience.2013.03.030 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

