MAP3K4 kinase activity dependent control of mouse gonadal sex determination†
- PMID: 33912929
- PMCID: PMC8660160
- DOI: 10.1093/biolre/ioab083
MAP3K4 kinase activity dependent control of mouse gonadal sex determination†
Abstract
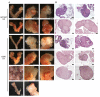
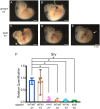
Sex determination requires the commitment of bipotential gonads to either a testis or an ovarian fate. Gene deletion of the kinase Map3k4 results in gonadal sex reversal in XY mice, and transgenic re-expression of Map3k4 rescues the sex reversal phenotype. Map3k4 encodes a large, multi-functional protein possessing a kinase domain and several, additional protein-protein interaction domains. Although MAP3K4 plays a critical role in male gonadal sex determination, it is unknown if the kinase activity of MAP3K4 is required. Here, we use mice expressing full-length, kinase-inactive MAP3K4 from the endogenous Map3k4 locus to examine the requirement of MAP3K4 kinase activity in sex determination. Although homozygous kinase-inactivation of MAP3K4 (Map3k4KI/KI) is lethal, a small fraction survive to adulthood. We show Map3k4KI/KI adults exhibit a 4:1 female-biased sex ratio. Many adult Map3k4KI/KI phenotypic females have a Y chromosome. XY Map3k4KI/KI adults with sex reversal display female mating behavior, but do not give rise to offspring. Reproductive organs are overtly female, but there is a broad spectrum of ovarian phenotypes, including ovarian absence, primitive ovaries, reduced ovarian size, and ovaries having follicles in all stages of development. Further, XY Map3k4KI/KI adults are smaller than either male or female Map3k4WT/WT mice. Examination of the critical stage of gonadal sex determination at E11.5 shows that loss of MAP3K4 kinase activity results in the loss of Sry expression in XY Map3k4KI/KI embryos, indicating embryonic male gonadal sex reversal. Together, these findings demonstrate the essential role for kinase activity of MAP3K4 in male gonadal sex determination.
Keywords: MAP2K3/6; MAP3K4; SRY; gonadal sex determination; kinase activity; mitogen-activated protein kinase kinase kinase 4; p38 MAPK.
© The Author(s) 2021. Published by Oxford University Press on behalf of Society for the Study of Reproduction. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures



References
-
- Carre GA, Greenfield A. Characterising novel pathways in testis determination using mouse genetics. Sexual Dev 2014; 8:199–207. - PubMed
-
- Stevant I, Nef S. Genetic control of gonadal sex determination and development. Trends Genet 2019; 35:346–358. - PubMed
-
- Karl J, Capel B. Sertoli cells of the mouse testis originate from the coelomic epithelium. Dev Biol 1998; 203:323–333. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

