Multiple Myeloma Patients Undergoing Carfilzomib: Development and Validation of a Risk Score for Cardiovascular Adverse Events Prediction
- PMID: 33915804
- PMCID: PMC8036868
- DOI: 10.3390/cancers13071631
Multiple Myeloma Patients Undergoing Carfilzomib: Development and Validation of a Risk Score for Cardiovascular Adverse Events Prediction
Abstract
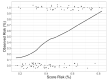
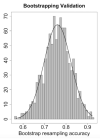
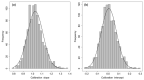
Cardiovascular adverse events (CVAEs) are linked to Carfilzomib (CFZ) therapy in multiple myeloma (MM); however, no validated protocols on cardiovascular risk assessment are available. In this prospective study, the effectiveness of the European Myeloma Network protocol (EMN) in cardiovascular risk assessment was investigated, identifying major predictors of CVAEs. From January 2015 to March 2020, 116 MM patients who had indication for CFZ therapy underwent a baseline evaluation (including blood pressure measurements, echocardiography and arterial stiffness estimation) and were prospectively followed. The median age was 64.53 ± 8.42 years old, 56% male. Five baseline independent predictors of CVAEs were identified: office systolic blood pressure, 24-h blood pressure variability, left ventricular hypertrophy, pulse wave velocity value and global longitudinal strain. The resulting 'CVAEs risk score' distinguished a low- and a high-risk group, obtaining a negative predicting value for the high-risk group of 90%. 52 patients (44.9%) experienced one or more CVAEs: 17 (14.7%) had major and 45 (38.7%) had hypertension-related events. In conclusion, CVAEs are frequent and a specific management protocol is crucial. The EMN protocol and the risk score proved to be useful to estimate the baseline risk for CVAEs during CFZ therapy, allowing the identification of higher-risk patients.
Keywords: arterial hypertension; cardiovascular adverse events; cardiovascular risk assessment; carfilzomib; echocardiography; global longitudinal strain; multiple myeloma; pulse wave velocity.
Conflict of interest statement
Alberto Milan received honoraria for advisory board from Amgen and Janssen. Sara Bringhen received honoraria from Bristol-Myers Squibb, Celgene, Amgen and Janssen; advisory boards for Amgen, Karyopharm, Janssen and Celgene; consultancy fees from Takeda and Janssen. Francesca Gay received honoraria from Amgen, Janseen, Celgene, BMS, Takeda, Abbvie; advisory boards for Amgen, Janseen, Celgene, BMS, Takeda, Abbvie; advisory adaptive for Roche Oncopeptides. The other authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
-
- Siegel D.S., Martin T., Wang M., Vij R., Jakubowiak A.J., Lonial S., Trudel S., Kukreti V., Bahlis N., Alsina M., et al. A phase 2 study of single-agent carfilzomib (PX-171-003-A1) in patients with relapsed and refractory multiple myeloma. Blood. 2012;120:2817–2825. doi: 10.1182/blood-2012-05-425934. - DOI - PMC - PubMed
-
- A Dimopoulos M., Moreau P., Palumbo A., Joshua D., Pour L., Hájek R., Facon T., Ludwig H., Oriol A., Goldschmidt H., et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): A randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17:27–38. doi: 10.1016/S1470-2045(15)00464-7. - DOI - PubMed
-
- Hájek R., Masszi T., Petrucci M.T., Palumbo A., Rosiñol L., Nagler A., Yong K.L., Oriol A., Minarik J., Pour L., et al. A randomized phase III study of carfilzomib vs low-dose corticosteroids with optional cyclophosphamide in relapsed and refractory multiple myeloma (FOCUS) Leukemia. 2017;31:107–114. doi: 10.1038/leu.2016.176. - DOI - PMC - PubMed
-
- Cornell R.F., Ky B., Weiss B.M., Dahm C.N., Gupta D.K., Du L., Carver J.R., Cohen A.D., Engelhardt B.G., Garfall A.L., et al. Prospective Study of Cardiac Events During Proteasome Inhibitor Therapy for Relapsed Multiple Myeloma. J. Clin. Oncol. 2019;37:1946–1955. doi: 10.1200/JCO.19.00231. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

