Nuclear Medicine Imaging in Neuroblastoma: Current Status and New Developments
- PMID: 33916640
- PMCID: PMC8066332
- DOI: 10.3390/jpm11040270
Nuclear Medicine Imaging in Neuroblastoma: Current Status and New Developments
Abstract
Neuroblastoma is the most common extracranial solid malignancy in children. At diagnosis, approximately 50% of patients present with metastatic disease. These patients are at high risk for refractory or recurrent disease, which conveys a very poor prognosis. During the past decades, nuclear medicine has been essential for the staging and response assessment of neuroblastoma. Currently, the standard nuclear imaging technique is meta-[123I]iodobenzylguanidine ([123I]mIBG) whole-body scintigraphy, usually combined with single-photon emission computed tomography with computed tomography (SPECT-CT). Nevertheless, 10% of neuroblastomas are mIBG non-avid and [123I]mIBG imaging has relatively low spatial resolution, resulting in limited sensitivity for smaller lesions. More accurate methods to assess full disease extent are needed in order to optimize treatment strategies. Advances in nuclear medicine have led to the introduction of radiotracers compatible for positron emission tomography (PET) imaging in neuroblastoma, such as [124I]mIBG, [18F]mFBG, [18F]FDG, [68Ga]Ga-DOTA peptides, [18F]F-DOPA, and [11C]mHED. PET has multiple advantages over SPECT, including a superior resolution and whole-body tomographic range. This article reviews the use, characteristics, diagnostic accuracy, advantages, and limitations of current and new tracers for nuclear medicine imaging in neuroblastoma.
Keywords: [11C]mHED; [123I]mIBG; [124I]mIBG; [18F]F-DOPA; [18F]FDG; [18F]mFBG; [68Ga]Ga-DOTA peptides; neuroblastoma; nuclear medicine; radionuclide imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
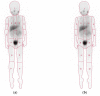
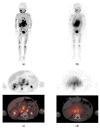
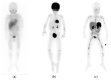
References
-
- Tas M.L., Reedijk A.M.J., Karim-Kos H.E., Kremer L.C.M., van de Ven C.P., Dierselhuis M.P., van Eijkelenburg N.K.A., van Grotel M., Kraal K.C.J.M., Peek A.M.L., et al. Neuroblastoma between 1990 and 2014 in the Netherlands: Increased incidence and improved survival of high-risk neuroblastoma. Eur. J. Cancer. 2020;124:47–55. doi: 10.1016/j.ejca.2019.09.025. - DOI - PubMed
-
- Morgenstern D.A., London W.B., Stephens D., Volchenboum S.L., Simon T., Nakagawara A., Shimada H., Schleiermacher G., Matthay K.K., Cohn S.L., et al. Prognostic significance of pattern and burden of metastatic disease in patients with stage 4 neuroblastoma: A study from the International Neuroblastoma Risk Group database. Eur. J. Cancer. 2016;65:1–10. doi: 10.1016/j.ejca.2016.06.005. - DOI - PubMed
-
- Monclair T., Brodeur G.M., Ambros P.F., Brisse H.J., Cecchetto G., Holmes K., Kaneko M., London W.B., Matthay K.K., Nuchtern J.G., et al. The International Neuroblastoma Risk Group (INRG) staging system: An INRG task force report. J. Clin. Oncol. 2009;27:298–303. doi: 10.1200/JCO.2008.16.6876. - DOI - PMC - PubMed
-
- Brodeur G.M., Pritchard J., Berthold F., Carlsen N.L.T., Castel V., Castleberry R.P., de Bernardi B., Evans A.E., Favrot M., Hedborg F., et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment. J. Clin. Oncol. 1993;11:1466–1477. doi: 10.1200/JCO.1993.11.8.1466. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

