Oxidative Stress-Induced Sirtuin1 Downregulation Correlates to HIF-1α, GLUT-1, and VEGF-A Upregulation in Th1 Autoimmune Hashimoto's Thyroiditis
- PMID: 33916948
- PMCID: PMC8067526
- DOI: 10.3390/ijms22083806
Oxidative Stress-Induced Sirtuin1 Downregulation Correlates to HIF-1α, GLUT-1, and VEGF-A Upregulation in Th1 Autoimmune Hashimoto's Thyroiditis
Abstract
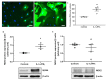
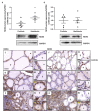
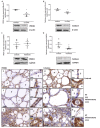
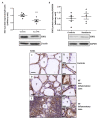
In Hashimoto's thyroiditis (HT), oxidative stress (OS) is driven by Th1 cytokines' response interfering with the normal function of thyrocytes. OS results from an imbalance between an excessive production of reactive oxygen species (ROS) and a lowering of antioxidant production. Moreover, OS has been shown to inhibit Sirtuin 1 (SIRT1), which is able to prevent hypoxia-inducible factor (HIF)-1α stabilization. The aims of this study were to determine the involvement of NADPH-oxidases (NOX), SIRT1, and HIF-1α in HT pathophysiology as well as the status of antioxidant proteins such as peroxiredoxin 1 (PRDX1), catalase, and superoxide dismutase 1 (SOD1). The protein expressions of NOX2, NOX4, antioxidant enzymes, SIRT1, and HIF-1α, as well as glucose transporter-1 (GLUT-1) and vascular endothelial growth factor A (VEGF-A), were analyzed by Western blot in primary cultures of human thyrocytes that were or were not incubated with Th1 cytokines. The same proteins were also analyzed by immunohistochemistry in thyroid samples from control and HT patients. In human thyrocytes incubated with Th1 cytokines, NOX4 expression was increased whereas antioxidants, such as PRDX1, catalase, and SOD1, were reduced. Th1 cytokines also induced a significant decrease of SIRT1 protein expression associated with an upregulation of HIF-1α, GLUT-1, and VEGF-A proteins. With the exception of PRDX1 and SOD1, similar results were obtained in HT thyroids. OS due to an increase of ROS produced by NOX4 and a loss of antioxidant defenses (PRDX1, catalase, SOD1) correlates to a reduction of SIRT1 and an upregulation of HIF 1α, GLUT-1, and VEGF-A. Our study placed SIRT1 as a key regulator of OS and we, therefore, believe it could be considered as a potential therapeutic target in HT.
Keywords: HIF-1α; Hashimoto’s thyroiditis; NOX4; Sirtuin1; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest. Authors have nothing to disclose. No competing financial interests exist.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

