Moderately Inducing Autophagy Reduces Tertiary Brain Injury after Perinatal Hypoxia-Ischemia
- PMID: 33919804
- PMCID: PMC8070811
- DOI: 10.3390/cells10040898
Moderately Inducing Autophagy Reduces Tertiary Brain Injury after Perinatal Hypoxia-Ischemia
Abstract
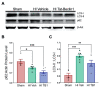
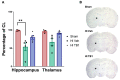
Recent studies of cerebral hypoxia-ischemia (HI) have highlighted slowly progressive neurodegeneration whose mechanisms remain elusive, but if blocked, could considerably improve long-term neurological function. We previously established that the cytokine transforming growth factor (TGF)β1 is highly elevated following HI and that delivering an antagonist for TGFβ receptor activin-like kinase 5 (ALK5)-SB505124-three days after injury in a rat model of moderate pre-term HI significantly preserved the structural integrity of the thalamus and hippocampus as well as neurological functions associated with those brain structures. To elucidate the mechanism whereby ALK5 inhibition reduces cell death, we assessed levels of autophagy markers in neurons and found that SB505124 increased numbers of autophagosomes and levels of lipidated light chain 3 (LC3), a key protein known to mediate autophagy. However, those studies did not determine whether (1) SB was acting directly on the CNS and (2) whether directly inducing autophagy could decrease cell death and improve outcome. Here we show that administering an ALK5 antagonist three days after HI reduced actively apoptotic cells by ~90% when assessed one week after injury. Ex vivo studies using the lysosomal inhibitor chloroquine confirmed that SB505124 enhanced autophagy flux in the injured hemisphere, with a significant accumulation of the autophagic proteins LC3 and p62 in SB505124 + chloroquine treated brain slices. We independently activated autophagy using the stimulatory peptide Tat-Beclin1 to determine if enhanced autophagy is directly responsible for improved outcomes. Administering Tat-Beclin1 starting three days after injury preserved the structural integrity of the hippocampus and thalamus with improved sensorimotor function. These data support the conclusion that intervening at this phase of injury represents a window of opportunity where stimulating autophagy is beneficial.
Keywords: autophagy; cell death; encephalopathy; neuroprotection; premature birth.
Conflict of interest statement
The authors declare no conflict of interests.
Figures



References
-
- Lloyd-Jones D., Adams R., Carnethon M., de Simone G., Ferguson T.B., Flegal K., Ford E., Furie K., Go A., Greenlund K., et al. Heart disease and stroke statistics—2009 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2009;119:480–486. doi: 10.1161/CIRCULATIONAHA.108.191259. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

