Detection of Sub-Nanomolar Concentration of Trypsin by Thickness-Shear Mode Acoustic Biosensor and Spectrophotometry
- PMID: 33920444
- PMCID: PMC8070231
- DOI: 10.3390/bios11040117
Detection of Sub-Nanomolar Concentration of Trypsin by Thickness-Shear Mode Acoustic Biosensor and Spectrophotometry
Abstract
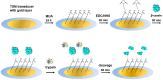
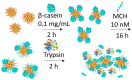
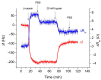
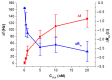
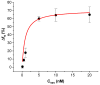
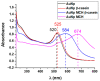
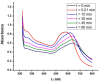
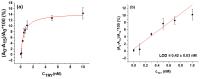
The determination of protease activity is very important for disease diagnosis, drug development, and quality and safety assurance for dairy products. Therefore, the development of low-cost and sensitive methods for assessing protease activity is crucial. We report two approaches for monitoring protease activity: in a volume and at surface, via colorimetric and acoustic wave-based biosensors operated in the thickness-shear mode (TSM), respectively. The TSM sensor was based on a β-casein substrate immobilized on a piezoelectric quartz crystal transducer. After an enzymatic reaction with trypsin, it cleaved the surface-bound β-casein, which increased the resonant frequency of the crystal. The limit of detection (LOD) was 0.48 ± 0.08 nM. A label-free colorimetric assay for trypsin detection has also been performed using β-casein and 6-mercaptohexanol (MCH) functionalized gold nanoparticles (AuNPs/MCH-β-casein). Due to the trypsin cleavage of β-casein, the gold nanoparticles lost shelter, and MCH increased the attractive force between the modified AuNPs. Consequently, AuNPs aggregated, and the red shift of the absorption spectra was observed. Spectrophotometric assay enabled an LOD of 0.42 ± 0.03 nM. The Michaelis-Menten constant, KM, for reverse enzyme reaction has also been estimated by both methods. This value for the colorimetric assay (0.56 ± 0.10 nM) is lower in comparison with those for the TSM sensor (0.92 ± 0.44 nM). This is likely due to the better access of the trypsin to the β-casein substrate at the surface of AuNPs in comparison with those at the TSM transducer.
Keywords: AuNPs; acoustic wave biosensor; colorimetric assay; trypsin; β-casein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Analysis of trypsin activity at β-casein layers formed on hydrophobic surfaces using a multiharmonic acoustic method.Analyst. 2022 Jan 31;147(3):461-470. doi: 10.1039/d1an01800k. Analyst. 2022. PMID: 34989356
-
A fluorometric and colorimetric method for determination of trypsin by exploiting the gold nanocluster-induced aggregation of hemoglobin-coated gold nanoparticles.Mikrochim Acta. 2019 Apr 8;186(5):272. doi: 10.1007/s00604-019-3380-2. Mikrochim Acta. 2019. PMID: 30963286
-
An optical biosensing platform for proteinase activity using gold nanoparticles.Biomaterials. 2010 Aug;31(23):6087-95. doi: 10.1016/j.biomaterials.2010.04.026. Epub 2010 May 14. Biomaterials. 2010. PMID: 20471084
-
Advances in Analysis of Milk Proteases Activity at Surfaces and in a Volume by Acoustic Methods.Sensors (Basel). 2020 Sep 29;20(19):5594. doi: 10.3390/s20195594. Sensors (Basel). 2020. PMID: 33003538 Free PMC article. Review.
-
A colorimetric aptasensor based on gold nanoparticles for detection of microbial toxins: an alternative approach to conventional methods.Anal Bioanal Chem. 2022 Oct;414(24):7103-7122. doi: 10.1007/s00216-022-04227-9. Epub 2022 Jul 29. Anal Bioanal Chem. 2022. PMID: 35902394 Review.
Cited by
-
Detection of Oenological Polyphenols via QCM-D Measurements.Nanomaterials (Basel). 2022 Jan 4;12(1):166. doi: 10.3390/nano12010166. Nanomaterials (Basel). 2022. PMID: 35010116 Free PMC article.
-
Application of Shear Horizontal Surface Acoustic Wave (SH-SAW) Immunosensor in Point-of-Care Diagnosis.Biosensors (Basel). 2023 Jun 1;13(6):605. doi: 10.3390/bios13060605. Biosensors (Basel). 2023. PMID: 37366970 Free PMC article. Review.
-
New Insights on Plasmin Long Term Stability and the Mechanism of Its Activity Inhibition Analyzed by Quartz Crystal Microbalance.Micromachines (Basel). 2021 Dec 29;13(1):55. doi: 10.3390/mi13010055. Micromachines (Basel). 2021. PMID: 35056220 Free PMC article.
-
Design and Functionality of Trypsin-Triggered, Expandable Bovine Serum Albumin-Polyethylene Glycol Diacrylate Hydrogel Actuators.Small Sci. 2024 Jul 21;4(10):2400214. doi: 10.1002/smsc.202400214. eCollection 2024 Oct. Small Sci. 2024. PMID: 40212243 Free PMC article.
-
From Enzymatic Dopamine Biosensors to OECT Biosensors of Dopamine.Biosensors (Basel). 2023 Aug 11;13(8):806. doi: 10.3390/bios13080806. Biosensors (Basel). 2023. PMID: 37622892 Free PMC article. Review.
References
-
- Glantz M., Rosenlow M., Lindmark-Månsson H., Johansen L.B., Hartmann J., Hojer A., Waak E., Lofgren R., Saeden K.H., Svensson S., et al. Impact of protease and lipase activities on quality of Swedish raw milk. Int. Dairy J. 2020;107:104724. doi: 10.1016/j.idairyj.2020.104724. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

