At What Age Could Screening for Familial Retinoblastoma Be Discontinued? A Systematic Review
- PMID: 33920538
- PMCID: PMC8072927
- DOI: 10.3390/cancers13081942
At What Age Could Screening for Familial Retinoblastoma Be Discontinued? A Systematic Review
Abstract
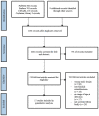
The aim of this systematic review is to assess the latest age at diagnosis for detection of familial retinoblastoma in order to evaluate at what age screening of at-risk children could be discontinued. Extended screening beyond this age would result in unnecessary patient burden and costs. However, discontinuing screening prematurely would have the adverse effect of missing tumors. We performed a literature search (PubMed, Embase, CINAHL and the Cochrane Library) up until February of 2021 and systematically included studies where patients had a family history of retinoblastoma, a known age at diagnosis, and who were ophthalmologically screened for retinoblastoma from birth. A total of 176 familial retinoblastoma patients from 17 studies were included in this review. Based on 48 months of age being the latest age of diagnosis, ophthalmological screening for familial retinoblastoma could safely be discontinued at age four years.
Keywords: age at diagnosis; familial retinoblastoma; screening.
Conflict of interest statement
Authors declare no conflict of interest.
Figures
References
-
- Munier F.L., Beck-Popovic M., Chantada G.L., Cobrinik D., Kivela T.T., Lohmann D., Maeder P., Moll A.C., Carcaboso A.M., Moulin A., et al. Conservative management of retinoblastoma: Challenging orthodoxy without compromising the state of metastatic grace. “Alive, with good vision and no comorbidity”. Prog. Retin. Eye Res. 2019;73:100764. doi: 10.1016/j.preteyeres.2019.05.005. - DOI - PubMed
-
- Rothschild P.R., Levy D., Savignoni A., Lumbroso-Le Rouic L., Aerts I., Gauthier-Villars M., Esteve M., Bours D., Desjardins L., Doz F., et al. Familial retinoblastoma: Fundus screening schedule impact and guideline proposal. A retrospective study. Eye. 2011;25:55–61. doi: 10.1038/eye.2011.198. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources