Treatment Efficacy with a Novel Hyaluronic Acid-Based Hydrogel for Osteoarthritis of the Knee
- PMID: 33920879
- PMCID: PMC8071312
- DOI: 10.3390/jpm11040303
Treatment Efficacy with a Novel Hyaluronic Acid-Based Hydrogel for Osteoarthritis of the Knee
Abstract
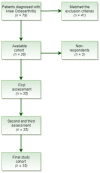
Background: Prior trials investigating the treatment of symptomatic osteoarthritis (OA) with hyaluronic-acid-derived products injections have provided optimistic results. The study was directed to assess the effectiveness of an innovative hyaluronic-acid-based hydrogel (Hymovis®) in the treatment of symptomatic knee OA. Methods: A prospective, single-center, clinical trial was performed. Thirty-five patients with degenerative knee OA were included. Inclusion criteria were: age between 45-80, radiographic Kellgren grade II or III osteoarthritis, minimum 35 mm score on the Visual Analogue Scale (VAS), pain for at least 6 months and agreement to participate in the study. Patients received two injections at a one-week interval. The evaluator assessed the patients using the Western Ontario and McMaster University Osteoarthritis Index (WOMAC) and VAS. Evaluation was performed before, at 2 and 6 months after the injections. Results: A significant improvement on the WOMAC Index pain subscale was observed at 6 months after the injection. At two months, pain subscale score decreased from 10.34 to 9.34. At six months, a significant decrement in pain parameters compared to baseline was observed (from 10.34 to 7.72; p = 0.0004). Median points on VAS significantly ameliorated after 6 months (from 74.2 to 57.3 cm; p < 0.0001). Regarding physical function, a statistically significant difference compared to baseline was observed at the end of the study (from 29.74 to 25.18; p = 0.0025). WOMAC Index stiffness component did not differ from baseline at any time during follow-up. Conclusions: Pain relief installed with a delayed on-set but had a prolonged duration. The novel hyaluronic acid-based hydrogel (Hymovis®) had effective results, particularly after six months post-injections and offers a therapeutic advancement in the treatment of moderate to severe osteoarthritis.
Keywords: hyaluronic acid; intra-articular injection; osteoarthritis.
Conflict of interest statement
The authors declare no conflict of interest declared in this section. The authors report no involvement in the research by the sponsor that could have influenced the outcome of this work.
Figures


References
-
- Vos T., Flaxman A.D., Naghavi M., Lozano R., Michaud C., Ezzati M., Shibuya K., Salomon J.A., Abdalla S., Aboyans V., et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet. 2012;380:2163–2196. doi: 10.1016/S0140-6736(12)61729-2. - DOI - PMC - PubMed
-
- McAlindon T.E., Bannuru R.R., Sullivan M.C., Arden N.K., Berenbaum F., Bierma-Zeinstra S.M., Hawker G.A., Henrotin Y., Hunter D.J., Kawaguchi H. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014;22:363–388. doi: 10.1016/j.joca.2014.01.003. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

