Structure and Activities of the NS1 Influenza Protein and Progress in the Development of Small-Molecule Drugs
- PMID: 33921888
- PMCID: PMC8074201
- DOI: 10.3390/ijms22084242
Structure and Activities of the NS1 Influenza Protein and Progress in the Development of Small-Molecule Drugs
Abstract
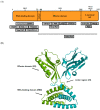
The influenza virus causes human disease on a global scale and significant morbidity and mortality. The existing vaccination regime remains vulnerable to antigenic drift, and more seriously, a small number of viral mutations could lead to drug resistance. Therefore, the development of a new additional therapeutic small molecule-based anti-influenza virus is urgently required. The NS1 influenza gene plays a pivotal role in the suppression of host antiviral responses, especially by inhibiting interferon (IFN) production and the activities of antiviral proteins, such as dsRNA-dependent serine/threonine-protein kinase R (PKR) and 2'-5'-oligoadenylate synthetase (OAS)/RNase L. NS1 also modulates important aspects of viral RNA replication, viral protein synthesis, and virus replication cycle. Taken together, small molecules that target NS1 are believed to offer a means of developing new anti-influenza drugs.
Keywords: NS1; drug; influenza virus A; inhibitor; small molecule.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
The influenza virus NS1 protein as a therapeutic target.Antiviral Res. 2013 Sep;99(3):409-16. doi: 10.1016/j.antiviral.2013.06.005. Epub 2013 Jun 21. Antiviral Res. 2013. PMID: 23796981 Free PMC article. Review.
-
The multifunctional NS1 protein of influenza A viruses.J Gen Virol. 2008 Oct;89(Pt 10):2359-2376. doi: 10.1099/vir.0.2008/004606-0. J Gen Virol. 2008. PMID: 18796704 Review.
-
The Short Form of the Zinc Finger Antiviral Protein Inhibits Influenza A Virus Protein Expression and Is Antagonized by the Virus-Encoded NS1.J Virol. 2017 Jan 3;91(2):e01909-16. doi: 10.1128/JVI.01909-16. Print 2017 Jan 15. J Virol. 2017. PMID: 27807230 Free PMC article.
-
NF90 is a novel influenza A virus NS1-interacting protein that antagonizes the inhibitory role of NS1 on PKR phosphorylation.FEBS Lett. 2016 Aug;590(16):2797-810. doi: 10.1002/1873-3468.12311. Epub 2016 Aug 5. FEBS Lett. 2016. PMID: 27423063
-
Influenza A Virus Virulence Depends on Two Amino Acids in the N-Terminal Domain of Its NS1 Protein To Facilitate Inhibition of the RNA-Dependent Protein Kinase PKR.J Virol. 2017 Apr 28;91(10):e00198-17. doi: 10.1128/JVI.00198-17. Print 2017 May 15. J Virol. 2017. PMID: 28250123 Free PMC article.
Cited by
-
The applications of live attenuated influenza a virus with modified NS1 gene.Mol Ther Nucleic Acids. 2025 Feb 1;36(1):102471. doi: 10.1016/j.omtn.2025.102471. eCollection 2025 Mar 11. Mol Ther Nucleic Acids. 2025. PMID: 40046952 Free PMC article. Review.
-
An overview of influenza A virus genes, protein functions, and replication cycle highlighting important updates.Virus Genes. 2022 Aug;58(4):255-269. doi: 10.1007/s11262-022-01904-w. Epub 2022 Apr 26. Virus Genes. 2022. PMID: 35471490 Review.
-
Antiviral responses versus virus-induced cellular shutoff: a game of thrones between influenza A virus NS1 and SARS-CoV-2 Nsp1.Front Cell Infect Microbiol. 2024 Feb 5;14:1357866. doi: 10.3389/fcimb.2024.1357866. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 38375361 Free PMC article. Review.
-
The Contribution of Viral Proteins to the Synergy of Influenza and Bacterial Co-Infection.Viruses. 2022 May 16;14(5):1064. doi: 10.3390/v14051064. Viruses. 2022. PMID: 35632805 Free PMC article. Review.
-
Interferon Control of Human Coronavirus Infection and Viral Evasion: Mechanistic Insights and Implications for Antiviral Drug and Vaccine Development.J Mol Biol. 2022 Mar 30;434(6):167438. doi: 10.1016/j.jmb.2021.167438. Epub 2022 Jan 3. J Mol Biol. 2022. PMID: 34990653 Free PMC article. Review.
References
-
- Shaw M., Palese P. Orthomyxoviridae: The viruses and their replication. Fields Virol. 2007;5:1647–1689.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

