Chagas Cardiomyopathy: From Romaña Sign to Heart Failure and Sudden Cardiac Death
- PMID: 33922366
- PMCID: PMC8145478
- DOI: 10.3390/pathogens10050505
Chagas Cardiomyopathy: From Romaña Sign to Heart Failure and Sudden Cardiac Death
Abstract
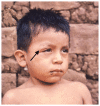
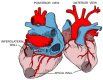
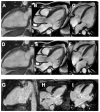
Despite nearly a century of research and accounting for the highest disease burden of any parasitic disease in the Western Hemisphere, Chagas disease (CD) is still a challenging diagnosis, primarily due to its poor recognition outside of Latin America. Although initially considered endemic to Central and South America, globalization, urbanization, and increased migration have spread the disease worldwide in the last few years, making it a significant public health threat. The international medical community's apparent lack of interest in this disease that was previously thought to be geographically restricted has delayed research on the complex host-parasite relationship that determines myocardial involvement and its differential behavior from other forms of cardiomyopathy, particularly regarding treatment strategies. Multiple cellular and molecular mechanisms that contribute to degenerative, inflammatory, and fibrotic myocardial responses have been identified and warrant further research to expand the therapeutic arsenal and impact the high burden attributed to CD. Altogether, cardiac dysautonomia, microvascular disturbances, parasite-mediated myocardial damage, and chronic immune-mediated injury are responsible for the disease's clinical manifestations, ranging from asymptomatic disease to severe cardiac and gastrointestinal involvement. It is crucial for healthcare workers to better understand CD transmission and disease dynamics, including its behavior on both its acute and chronic phases, to make adequate and evidence-based decisions regarding the disease. This review aims to summarize the most recent information on the epidemiology, pathogenesis, clinical presentation, diagnosis, screening, and treatment of CD, emphasizing on Chagasic cardiomyopathy's (Ch-CMP) clinical presentation and pathobiological mechanisms leading to sudden cardiac death.
Keywords: Ch-CMP/Chagas cardiomyopathy; Chagas disease; heart failure; sudden cardiac death; trypanosoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

