Synthesis of Eco-Friendly Biopolymer, Alginate-Chitosan Composite to Adsorb the Heavy Metals, Cd(II) and Pb(II) from Contaminated Effluents
- PMID: 33923314
- PMCID: PMC8123203
- DOI: 10.3390/ma14092189
Synthesis of Eco-Friendly Biopolymer, Alginate-Chitosan Composite to Adsorb the Heavy Metals, Cd(II) and Pb(II) from Contaminated Effluents
Abstract
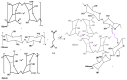
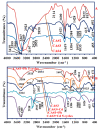
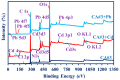
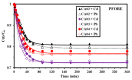
Efficient removal of Cd(II) and Pb(II) from contaminated water is considered a fundamental point of view. Synthetic hydrogel biopolymers based on chitosan and alginate (cost-effective and eco-friendly) were successfully designed and characterized by highly efficient removal contaminants. The sorbents are characterized by FTIR, SEM-EDX, TGA, XPS analyses and textural properties which are qualified by N2 adsorption. The sorption properties are firstly investigated by the effect of pH, sorption isotherms, uptake kinetics, and selectivity from multi-metal solution with equi-molar concentration. The sorbent with 1:3 ratios (of chitosan and alginate respectively) is the most effective for metal removal (i.e., 0.81 mmol Cd g-1 and 0.41 mmol Pb g-1). Langmuir and Sip's models fitted better the adsorption isotherms compared to the Freundlich model. Uptake kinetics was well fitted by pseudo-first-order rate equation, while the saturation was achieved within 40 min. The sorbent shows good reproducibility through duplicate the experiments with negligible decreasing efficiency (>2.5%). The sorbent was applied for water treatment on samples collected from the industrial area (i.e., 653 and 203 times over the MCL for Cd(II) and Pb(II) respectively according to WHO). The concentration of Cd and Pb was drastically decreased in the effluents as pH increased with removal efficiency up to 99% for both elements at pH 5.8 and SD equivalent 1 g L-1 for 5 h.
Keywords: cadmium and lead contamination; contaminated water treatment; cost-effective biopolymers; eco-friendly sorbent.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- WHO . Guidelines for Drinking-Water Quality. 4th ed. World Health Organization; Geneva, Switzerland: 2011. p. 541.
-
- Selim M.T., Salem S.S., Mohamed A.A., El-Gamal M.S., Awad M.F., Fouda A. Biological treatment of real textile effluent using Aspergillus flavus and Fusarium oxysporium and their consortium along with the evaluation of their phytotoxicity. J. Fungi. 2021;7:193. doi: 10.3390/jof7030193. - DOI - PMC - PubMed
-
- EU . Document 31998L0083. European Union; Brussels, Belgium: 1998. Council Directive 98/83/EC of 3 November 1998 on the quality of water intented for human consumption.
-
- USEPA . The Drinking Water Standards and Health Advisories. Volume 822-S-12-001. Office of Water, U.S. Environmental Protection Agency; Washington, DC, USA: 2012. pp. 1–12.
LinkOut - more resources
Full Text Sources
Miscellaneous

