Genes Implicated in Familial Parkinson's Disease Provide a Dual Picture of Nigral Dopaminergic Neurodegeneration with Mitochondria Taking Center Stage
- PMID: 33924963
- PMCID: PMC8124903
- DOI: 10.3390/ijms22094643
Genes Implicated in Familial Parkinson's Disease Provide a Dual Picture of Nigral Dopaminergic Neurodegeneration with Mitochondria Taking Center Stage
Abstract
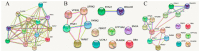
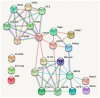
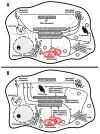
The mechanism of nigral dopaminergic neuronal degeneration in Parkinson's disease (PD) is unknown. One of the pathological characteristics of the disease is the deposition of α-synuclein (α-syn) that occurs in the brain from both familial and sporadic PD patients. This paper constitutes a narrative review that takes advantage of information related to genes (SNCA, LRRK2, GBA, UCHL1, VPS35, PRKN, PINK1, ATP13A2, PLA2G6, DNAJC6, SYNJ1, DJ-1/PARK7 and FBXO7) involved in familial cases of Parkinson's disease (PD) to explore their usefulness in deciphering the origin of dopaminergic denervation in many types of PD. Direct or functional interactions between genes or gene products are evaluated using the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) database. The rationale is to propose a map of the interactions between SNCA, the gene encoding for α-syn that aggregates in PD, and other genes, the mutations of which lead to early-onset PD. The map contrasts with the findings obtained using animal models that are the knockout of one of those genes or that express the mutated human gene. From combining in silico data from STRING-based assays with in vitro and in vivo data in transgenic animals, two likely mechanisms appeared: (i) the processing of native α-syn is altered due to the mutation of genes involved in vesicular trafficking and protein processing, or (ii) α-syn mutants alter the mechanisms necessary for the correct vesicular trafficking and protein processing. Mitochondria are a common denominator since both mechanisms require extra energy production, and the energy for the survival of neurons is obtained mainly from the complete oxidation of glucose. Dopamine itself can result in an additional burden to the mitochondria of dopaminergic neurons because its handling produces free radicals. Drugs acting on G protein-coupled receptors (GPCRs) in the mitochondria of neurons may hopefully end up targeting those receptors to reduce oxidative burden and increase mitochondrial performance. In summary, the analysis of the data of genes related to familial PD provides relevant information on the etiology of sporadic cases and might suggest new therapeutic approaches.
Keywords: Lewy bodies; early-onset Parkinson’s disease; familial Parkinson’s disease; mitochondria; mitophagy; synuclein aggregation; vesicular transport.
Conflict of interest statement
Authors declare no conflict of interests.
Figures
Similar articles
-
Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse.Free Radic Biol Med. 2013 Sep;62:186-201. doi: 10.1016/j.freeradbiomed.2013.05.042. Epub 2013 Jun 3. Free Radic Biol Med. 2013. PMID: 23743292 Review.
-
Molecular chaperones and Parkinson's disease.Neurobiol Dis. 2021 Dec;160:105527. doi: 10.1016/j.nbd.2021.105527. Epub 2021 Oct 7. Neurobiol Dis. 2021. PMID: 34626793 Review.
-
The deglycase activity of DJ-1 mitigates α-synuclein glycation and aggregation in dopaminergic cells: Role of oxidative stress mediated downregulation of DJ-1 in Parkinson's disease.Free Radic Biol Med. 2019 May 1;135:28-37. doi: 10.1016/j.freeradbiomed.2019.02.014. Epub 2019 Feb 20. Free Radic Biol Med. 2019. PMID: 30796974
-
alpha -Synucleinopathy and selective dopaminergic neuron loss in a rat lentiviral-based model of Parkinson's disease.Proc Natl Acad Sci U S A. 2002 Aug 6;99(16):10813-8. doi: 10.1073/pnas.152339799. Epub 2002 Jul 16. Proc Natl Acad Sci U S A. 2002. PMID: 12122208 Free PMC article.
-
Outlook of SNCA (α-synuclein) transgenic fly models in delineating the sequel of mitochondrial dysfunction in Parkinson's disease.Brain Res. 2025 Apr 1;1852:149505. doi: 10.1016/j.brainres.2025.149505. Epub 2025 Feb 13. Brain Res. 2025. PMID: 39954798 Review.
Cited by
-
Cannabinoid regulation of angiotensin II-induced calcium signaling in striatal neurons.NPJ Parkinsons Dis. 2024 Nov 15;10(1):220. doi: 10.1038/s41531-024-00827-7. NPJ Parkinsons Dis. 2024. PMID: 39548112 Free PMC article.
-
Zebrafish metabolomics: a comprehensive approach to understanding health and disease.Funct Integr Genomics. 2025 May 28;25(1):110. doi: 10.1007/s10142-025-01621-1. Funct Integr Genomics. 2025. PMID: 40425969 Review.
-
Alpha-Synuclein Aggregation in Parkinson's Disease.Front Med (Lausanne). 2021 Oct 18;8:736978. doi: 10.3389/fmed.2021.736978. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34733860 Free PMC article. Review.
-
Neuroprotection against Aminochrome Neurotoxicity: Glutathione Transferase M2-2 and DT-Diaphorase.Antioxidants (Basel). 2022 Jan 31;11(2):296. doi: 10.3390/antiox11020296. Antioxidants (Basel). 2022. PMID: 35204179 Free PMC article. Review.
-
Robustness of the Krebs Cycle under Physiological Conditions and in Cancer: New Clues for Evaluating Metabolism-Modifying Drug Therapies.Biomedicines. 2022 May 22;10(5):1199. doi: 10.3390/biomedicines10051199. Biomedicines. 2022. PMID: 35625935 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

