Advances in Twin-Screw Granulation Processing
- PMID: 33925577
- PMCID: PMC8146340
- DOI: 10.3390/pharmaceutics13050624
Advances in Twin-Screw Granulation Processing
Abstract
Twin-screw granulation (TSG) is a pharmaceutical process that has gained increased interest from the pharmaceutical industry for its potential for the development of oral dosage forms. The technology has evolved rapidly due to the flexibility of the equipment design, the selection of the process variables and the wide range of processed materials. Most importantly, TSG offers the benefits of both batch and continuous manufacturing for pharmaceutical products, accompanied by excellent process control, high product quality which can be achieved through the implementation of Quality by Design (QbD) approaches and the integration of Process Analytical Tools (PAT). Here, we present basic concepts of the various twin-screw granulation techniques and present in detail their advantages and disadvantages. In addition, we discuss the detail of the instrumentation used for TSG and how the critical processing paraments (CPP) affect the critical quality attributes (CQA) of the produced granules. Finally, we present recent advances in TSG continuous manufacturing including the paradigms of modelling of continuous granulation process, QbD approaches coupled with PAT monitoring for granule optimization and process understanding.
Keywords: PAT tools; QbD; continuous processing; granulation mechanisms; twin-screw granulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






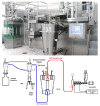


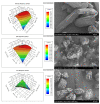
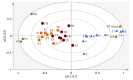




References
-
- Parikh D.M. Handbook of Pharmaceutical Granulation Technology. CRC Press; Boca Raton, FL, USA: 2016.
-
- Walker G.M. In: Chapter 4 Drum Granulation Processes. Granulation, Salman A.D., Hounslow M.J., Seville JPKBT-H of PT, editors. Elsevier Science B.V.; Radarweg, The Netherlands: 2007. pp. 219–254.
-
- Bolhuis G.K., de Waard H. Compaction properties of directly compressible materials. Pharm. Powder Compact. Technol. 2011;2:154.
-
- Aulton M.E., Taylor K.M.G. Aulton’s Pharmaceutics. 4th ed. Churchill Livingstone Elsevier; Edinburgh, UK: 2013. Pharmaceutical Preformulation.
-
- Aulton M.E. Pharmaceutics. The Science of Dosage Form Design. 2nd ed. Churchill Livingstone; Harcourt Publ Limited; London, UK: 2005.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

