Generative Models for Active Vision
- PMID: 33927605
- PMCID: PMC8076554
- DOI: 10.3389/fnbot.2021.651432
Generative Models for Active Vision
Abstract
The active visual system comprises the visual cortices, cerebral attention networks, and oculomotor system. While fascinating in its own right, it is also an important model for sensorimotor networks in general. A prominent approach to studying this system is active inference-which assumes the brain makes use of an internal (generative) model to predict proprioceptive and visual input. This approach treats action as ensuring sensations conform to predictions (i.e., by moving the eyes) and posits that visual percepts are the consequence of updating predictions to conform to sensations. Under active inference, the challenge is to identify the form of the generative model that makes these predictions-and thus directs behavior. In this paper, we provide an overview of the generative models that the brain must employ to engage in active vision. This means specifying the processes that explain retinal cell activity and proprioceptive information from oculomotor muscle fibers. In addition to the mechanics of the eyes and retina, these processes include our choices about where to move our eyes. These decisions rest upon beliefs about salient locations, or the potential for information gain and belief-updating. A key theme of this paper is the relationship between "looking" and "seeing" under the brain's implicit generative model of the visual world.
Keywords: Bayesian; active vision; attention; generative model; inference; oculomotion.
Copyright © 2021 Parr, Sajid, Da Costa, Mirza and Friston.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
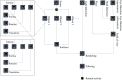


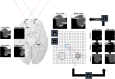
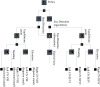

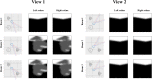
References
-
- Adler A. (1944). Disintegration and restoration of optic recognition in visual agnosia: analysis of a case. Arch. Neurol. Psychiatry 51, 243–259. 10.1001/archneurpsyc.1944.02290270032004 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

