Antibody conjugation and formulation
- PMID: 33928219
- PMCID: PMC7990145
- DOI: 10.1093/abt/tbz002
Antibody conjugation and formulation
Abstract
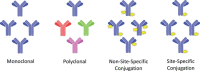
In an era where ultra-high antibody concentrations, high viscosities, low volumes, auto-injectors and long storage requirements are already complex problems with the current unconjugated monoclonal antibodies on the market, the formulation demands for antibody-drug conjugates (ADCs) are significant. Antibodies have historically been administered at relatively low concentrations through intravenous (IV) infusion due to their large size and the inability to formulate for oral delivery. Due to the high demands associated with IV infusion and the development of novel antibody targets and unique antibody conjugates, more accessible routes of administration such as intramuscular and subcutaneous are being explored. This review will summarize various site-specific and non-site-specific antibody conjugation techniques in the context of ADCs and the demands of formulation for high concentration clinical implementation.
Keywords: antibody-drug conjugates; conjugation; formulation; high concentration; site-specific.
© The Author(s) 2019. Published by Oxford University Press on behalf of Antibody Therapeutics.
Figures

References
-
- Dempke, WCM, Fenchel, K, Uciechowski, Pet al. . Second- and third-generation drugs for immuno-oncology treatment—the more the better? Eur J Cancer 2017; 74: 55–72. - PubMed
-
- Beck, A, Goetsch, L, Dumontet, Cet al. . Strategies and challenges for the next generation of antibody drug conjugates. Nat Rev Drug Discov 2017; 16: 315–37. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
