Clinicopathological features of tumor mutation burden, Epstein-Barr virus infection, microsatellite instability and PD-L1 status in Chinese patients with gastric cancer
- PMID: 33933102
- PMCID: PMC8088709
- DOI: 10.1186/s13000-021-01099-y
Clinicopathological features of tumor mutation burden, Epstein-Barr virus infection, microsatellite instability and PD-L1 status in Chinese patients with gastric cancer
Abstract
Objectives: Gastric cancer (GC) is the 4th most common type of cancer worldwide. Different GC subtypes have unique molecular features that may have different therapeutic methods. The aim of the present study was to investigate Epstein-Barr virus (EBV) infection, microsatellite instability (MSI) status, the expression of programmed death-ligand 1 (PD-L1) and gene mutations in GC patients.
Methods: The data of 2504 GC patients, who underwent curative gastrectomy with lymphadenectomy at Peking University Cancer Hospital between 2013 and 2018, were reviewed. We analyzed the clinicopathological factors associated with the immunohistochemistry (IHC) profiles of these patients, and genetic alterations were analyzed using next generation sequencing (NGS).
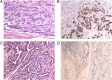
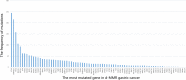
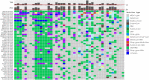
Results: Mismatch repair-deficient (d-MMR) GC patients were found to have a higher probability of expressing PD-L1 (p = 0.000, PD-L1 cutoff value = 1%). In addition, 4 and 6.9% of the 2504 gastric cancer patients were EBV-positive and d-MMR, respectively. The number of MLH1/PMS2-negative cases was 126 (6%), and the number of MSH2/MSH6-negative cases was 14 (0.9%). d-MMR status was associated with a intestinal group (p = 0.012), but not with tumor differentiation. Furthermore, MSI and d-MMR GC status (detected by NGS and IHC, respectively) were consistently high, and the rate of MSI was higher in patients with d-MMR GC. A number of genes associated with DNA damage repair were detected in GC patients with MSI, including POLE, ETV6, BRCA and RNF43. In patients with a high tumor mutation burden, the most significantly mutated genes were LRP1B (79.07%), ARID1A (74.42%), RNF43 (69.77%), ZFHX3 (65.12%), TP53 (58.14%), GANS (51.16%), BRCA2 (51.16%), PIK3CA (51.16%), NOTCH1 (51.16%), SMARCA4 (48.84%), ATR (46.51%), POLE (41.86%) and ATM (39.53%).
Conclusions: Using IHC and NGS, MSI status, protein expression, tumor mutation burden (TMB) and genetic alterations were identified in patients with GC, which provides a theoretical basis for the future clinical treatment of GC.
Keywords: EBV-encoded RNA; Gastric cancer; MSI; Next generation sequencing; PD-L1; TMB.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Clinicopathological and prognostic features of Epstein-Barr virus infection, microsatellite instability, and PD-L1 expression in gastric cancer.J Surg Oncol. 2018 Apr;117(5):829-839. doi: 10.1002/jso.25022. Epub 2018 Mar 13. J Surg Oncol. 2018. PMID: 29534305
-
[Comprehensive assessment of mismatch repair and microsatellite instability status in molecular classification of endometrial carcinoma].Zhonghua Fu Chan Ke Za Zhi. 2023 Oct 25;58(10):755-765. doi: 10.3760/cma.j.cn112141-20230711-00316. Zhonghua Fu Chan Ke Za Zhi. 2023. PMID: 37849256 Chinese.
-
Clinicopathological features of 22C3 PD-L1 expression with mismatch repair, Epstein-Barr virus status, and cancer genome alterations in metastatic gastric cancer.Gastric Cancer. 2019 Jan;22(1):69-76. doi: 10.1007/s10120-018-0843-9. Epub 2018 Jun 1. Gastric Cancer. 2019. PMID: 29859006
-
ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: a systematic review-based approach.Ann Oncol. 2019 Aug 1;30(8):1232-1243. doi: 10.1093/annonc/mdz116. Ann Oncol. 2019. PMID: 31056702
-
Epstein-Barr virus-associated gastric cancer: disease that requires special approach.Gastric Cancer. 2020 Nov;23(6):951-960. doi: 10.1007/s10120-020-01095-z. Epub 2020 Jun 8. Gastric Cancer. 2020. PMID: 32514646 Review.
Cited by
-
Cadonilimab in combination with chemotherapy for HER2-negative advanced gastric or gastroesophageal junction adenocarcinoma: a cost-effectiveness analysis.Front Pharmacol. 2025 Jul 29;16:1646818. doi: 10.3389/fphar.2025.1646818. eCollection 2025. Front Pharmacol. 2025. PMID: 40799822 Free PMC article.
-
Low incidence of microsatellite instability in gastric cancers and its association with the clinicopathological characteristics: a comparative study.Sci Rep. 2023 Dec 8;13(1):21743. doi: 10.1038/s41598-023-48157-7. Sci Rep. 2023. PMID: 38065969 Free PMC article.
-
Immunotherapy for diffuse gastric cancer: challenges and new avenues.NPJ Precis Oncol. 2025 Jul 21;9(1):247. doi: 10.1038/s41698-025-01052-y. NPJ Precis Oncol. 2025. PMID: 40691286 Free PMC article. Review.
-
LncRNA and its role in gastric cancer immunotherapy.Front Cell Dev Biol. 2023 Feb 16;11:1052942. doi: 10.3389/fcell.2023.1052942. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 36875764 Free PMC article. Review.
-
Heterogeneity and Adjuvant Therapeutic Approaches in MSI-H/dMMR Resectable Gastric Cancer: Emerging Trends in Immunotherapy.Ann Surg Oncol. 2023 Dec;30(13):8572-8587. doi: 10.1245/s10434-023-14103-0. Epub 2023 Sep 4. Ann Surg Oncol. 2023. PMID: 37667098 Free PMC article. Review.
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. 10.3322/caac.21590. Epub 2020 Jan 8. - PubMed
-
- Kim MY, Kruger AJ, Jeong JY, Kim J, Shin PK, Kim SY, Cho JY, Hahm KB, Hong SP. Combination therapy with a PI3K/mTOR dual inhibitor and Chloroquine enhances synergistic apoptotic cell death in Epstein-Barr virus-infected gastric Cancer cells. Mol Cell. 2019;42(6):448–459. doi: 10.14348/molcells.2019.2395. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

