A Comparative Analysis of Once-daily and Twice-daily Formulation of Tacrolimus in De Novo Kidney Transplant Recipients
- PMID: 33935537
- PMCID: PMC8085452
- DOI: 10.14744/SEMB.2020.71235
A Comparative Analysis of Once-daily and Twice-daily Formulation of Tacrolimus in De Novo Kidney Transplant Recipients
Abstract
Objectives: We aimed to compare the once-daily and twice-daily formulation of tacrolimus concerning the efficiency and effects on graft function in de novo kidney transplant patients.
Methods: Twenty once-daily (TAC-OD) and twenty twice-daily (TAC-BID) tacrolimus administrated de novo kidney recipients who had received initial immunosuppressive therapy according to protocols at our institution (0.2 mg/kg of tacrolimus combined with 1000 milligrams of steroid taper plus 720 mg of mycophenolate and with 2.5mg/kg anti-thymocyte globulin) assessed concerning demographics, drug doses and blood concentration, and graft function.
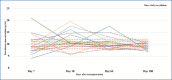
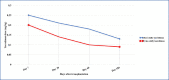
Results: The mean tacrolimus blood concentration measurements were higher in the TAC-OD group in the first sixty days after transplantation, and the TAC-OD group showed more blood concentration overshoots/fluctuations in the first 30 days of the treatment. The initial drug dose was significantly higher in the TAC-OD group than the TAC-BID group (p=0.04). There was no meaningful difference among groups according to graft function (creatinine measurements) (p>0.05).
Conclusion: Between de novo kidney recipients, the new TAC-OD formulation presents a similar short-term efficacy profile as TAC-BID. However, a higher daily dosage of TAC-OD is needed to achieve similar blood concentrations in the early postoperative period.
Keywords: Kidney transplantation; immunosuppression; prolonged-release tacrolimus; tacrolimus.
Copyright: © 2021 by The Medical Bulletin of Sisli Etfal Hospital.
Conflict of interest statement
Conflict of Interest: None declared.
Figures


References
-
- Banas B, Krämer BK, Krüger B, Kamar N, Undre N. Long-term kidney transplant outcomes:role of prolonged-release tacrolimus. Transplant Proc. 2020;52:102–10. - PubMed
-
- Danguilan RA, Lamban A, Pamugas GEP. Pilot study comparing the efficacy safety, convertibility and tacrolimus trough levels of twice-daily tacrolimus (prograf) to once-daily tacrolimus (advagraf) among standard-risk kidney transplant patients at the national kidney and transplant institute. Transplant Proc. 2019;51:2615–9. - PubMed
-
- Sukkha S, Suansanae T, Iamrahong P, Wiwattanathum P. Trough level and tacrolimus variability of early converted once-daily tacrolimus:1-Year Follow-up Study. Transplant Proc. 2020;52:775–9. - PubMed
-
- First MR. First clinical experience with the new once-daily formulation of tacrolimus. Ther Drug Monit. 2008;30:159–66. - PubMed
-
- First MR, Fitzsimmons WE. Modified release tacrolimus. Yonsei Med J. 2004;45:1127–31. - PubMed
LinkOut - more resources
Full Text Sources
