Development of an Advanced Multicellular Intestinal Model for Assessing Immunomodulatory Properties of Anti-Inflammatory Compounds
- PMID: 33935729
- PMCID: PMC8085553
- DOI: 10.3389/fphar.2021.639716
Development of an Advanced Multicellular Intestinal Model for Assessing Immunomodulatory Properties of Anti-Inflammatory Compounds
Abstract
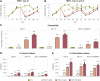
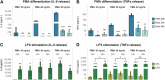
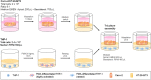
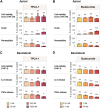
Intestinal inflammation is the collective term for immune system-mediated diseases of unknown, multifactorial etiology, with often complex interactions between genetic and environmental factors. To mechanistically investigate the effect of treatment with compounds possessing immunomodulating properties in the context of intestinal inflammation, we developed an immunocompetent in vitro triculture intestinal model consisting of a differentiated intestinal epithelial layer (Caco-2/HT29-MTX) and immunocompetent cells (differentiated THP-1). The triculture mimicked a healthy intestine with stable barrier integrity. Lipopolysaccharide treatment triggered a controlled and reversible inflammatory state, resulting in significant impairment of barrier integrity and release of pro-inflammatory cytokines and chemokines, which are known hallmarks of intestinal inflammation. Treatment with known anti-inflammatory reference compounds (TPCA-1 and budenoside) prevented the induction of an inflammatory state; the decreasing triculture responses to this treatment measured by cytokine release, transepithelial electric resistance (TEER), and epithelial layer permeability proved the suitability of the intestinal model for anti-inflammatory drug screening. Finally, selected tobacco alkaloids (nicotine and anatabine (R/S and S forms)) were tested in the in vitro triculture for their potential anti-inflammatory properties. Indeed, naturally occurring alkaloids, such as tobacco-derived alkaloids, have shown substantial anti-inflammatory effects in several in vitro and in vivo models of inflammation, gaining increasing interest. Similar to the anti-inflammatory reference compounds, one of the tobacco alkaloids under investigation partially prevented the decrease in the TEER and increase in permeability and reduced the release of pro-inflammatory cytokines and chemokines. Taken together, these data confirm that our in vitro model is suitable for screening potential anti-inflammatory compounds in the context of intestinal inflammation.
Keywords: anatabine; immune competent cells; in vitro co-culture; inflamed intestine; inflammatory bowel disease; intestinal inflammation; nicotine; tobacco alkaloids.
Copyright © 2021 Marescotti, Lo Sasso, Guerrera, Renggli, Ruiz Castro, Piault, Jaquet, Moine, Luettich, Frentzel, Peitsch and Hoeng.
Conflict of interest statement
All authors are employees of Philip Morris International R&D or had worked for Philip Morris International R&D under contractual agreements.
Figures


References
-
- Abreu M. T., Vora P., Faure E., Thomas L. S., Arnold E. T., Arditi M. (2001). Decreased Expression of Toll-like Receptor-4 and MD-2 Correlates with Intestinal Epithelial Cell Protection against Dysregulated Proinflammatory Gene Expression in Response to Bacterial Lipopolysaccharide. J. Immunol. 167 (3), 1609–1616. 10.4049/jimmunol.167.3.1609 - DOI - PubMed
-
- Alijevic O., McHugh D., Rufener L., Mazurov A., Hoeng J., Peitsch M. (2020). An Electrophysiological Characterization of Naturally Occurring Tobacco Alkaloids and Their Action on Human α4β2 and α7 Nicotinic Acetylcholine Receptors. Phytochemistry 170, 112187. 10.1016/j.phytochem.2019.112187 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

