Apoptosis induction by siRNA targeting integrin-β1 and regorafenib/DDAB-mPEG-PCL hybrid nanoparticles in regorafenib-resistant colon cancer cells
- PMID: 33948352
- PMCID: PMC8085858
Apoptosis induction by siRNA targeting integrin-β1 and regorafenib/DDAB-mPEG-PCL hybrid nanoparticles in regorafenib-resistant colon cancer cells
Abstract
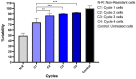
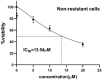
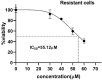
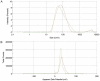
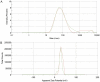
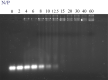
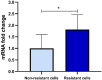
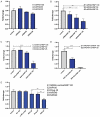
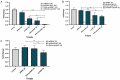
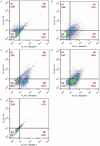
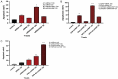
Colorectal cancer (CRC) is regarded as the third most common cancer worldwide. Although Regorafenib as a receptor tyrosine kinase inhibitor (RTKI) disrupts tumor growth and angiogenesis in metastatic CRC (mCRC) patients, drug resistance leads to poor prognosis and survival. Integrin-β1 overexpression has been proposed to be the major player in this regard. Herein, the Regorafenib-resistant human colon cancer cell line (SW-48) was induced, and the Integrin-β1 gene expression, as well as apoptosis, was assessed through the combination of small interfering RNA (siRNA) targeting Integrin-β1 and Regorafenib/Dimethyldioctadecylammonium bromide (DDAB)-methoxy poly (ethylene glycol) (mPEG)-poly-ε-caprolactone (PCL) hybrid nanoparticles (HNPs). In the current study, Regorafenib-resistant SW-48 cell line was generated in which the Regorafenib half-maximal inhibitory concentration (IC50) for non-resistant and resistant cells was 13.5±1.5 µM and 55.1±0.8 µM, respectively. The results of DLS also demonstrated that the size and the charge of the HNPs were equal to 66.56±0.5 nm and +29.5±1.2 mv, respectively. In addition, the Integrin-β1 gene expression was significantly higher in resistant cells than in non-resistant ones (P<0.05). The siRNA/HNP complexes in combination with Regorafenib/HNPs were accordingly identified as the most effective treatment to decrease the Integrin-β1 gene expression and to enhance the apoptosis rate in resistant cells (P<0.001). Overall, the study indicated that combination therapy using siRNA/HNP and Regorafenib/HNPs complex could down-regulate the Integrin-β1 gene expression and consequently trigger apoptosis, and this may potentially induce drug sensitivity.
Keywords: Colorectal cancer; SiRNA; apoptosis; integrin-β1; lipid-polymer hybrid nanoparticle; regorafenib.
AJCR Copyright © 2021.
Conflict of interest statement
None.
Figures
Similar articles
-
Combination of Quercetin or/and siRNA-loaded DDAB-mPEG-PCL hybrid nanoparticles reverse resistance to Regorafenib in colon cancer cells.BMC Complement Med Ther. 2022 Dec 27;22(1):340. doi: 10.1186/s12906-022-03787-8. BMC Complement Med Ther. 2022. PMID: 36575448 Free PMC article.
-
DDAB cationic lipid-mPEG, PCL copolymer hybrid nano-carrier synthesis and application for delivery of siRNA targeting IGF-1R into breast cancer cells.Clin Transl Oncol. 2021 Jun;23(6):1167-1178. doi: 10.1007/s12094-020-02507-3. Epub 2021 Jan 3. Clin Transl Oncol. 2021. PMID: 33389648
-
Dual silencing of integrin αvβ3 receptor and insulin-like growth factor 1 receptor using mPEG-PCL/DDAB hybrid nanoparticle loaded siRNA in breast cancer therapy: An in vitro study on MCF-7 cells.Int J Biol Macromol. 2025 Mar;294:139334. doi: 10.1016/j.ijbiomac.2024.139334. Epub 2024 Dec 30. Int J Biol Macromol. 2025. PMID: 39743068
-
Regorafenib: a novel multitargeted tyrosine kinase inhibitor for colorectal cancer and gastrointestinal stromal tumors.Ann Pharmacother. 2013 Dec;47(12):1685-96. doi: 10.1177/1060028013509792. Epub 2013 Nov 1. Ann Pharmacother. 2013. PMID: 24259629 Review.
-
Regorafenib: A Review in Metastatic Colorectal Cancer.Drugs. 2018 Jul;78(11):1133-1144. doi: 10.1007/s40265-018-0938-y. Drugs. 2018. PMID: 29943375 Review.
Cited by
-
Exploring Gene Therapy: The Next Generation of Colorectal Cancer Treatment.Curr Gene Ther. 2025;25(3):195-198. doi: 10.2174/0115665232326072240809061901. Curr Gene Ther. 2025. PMID: 40351072 No abstract available.
-
CircRNA_002178 as a ceRNA promotes the development of colorectal cancer by regulating miR-542-3p/CREB1.Am J Transl Res. 2021 Sep 15;13(9):10038-10055. eCollection 2021. Am J Transl Res. 2021. PMID: 34650680 Free PMC article.
-
Controlled-Release Nanosystems with a Dual Function of Targeted Therapy and Radiotherapy in Colorectal Cancer.Pharmaceutics. 2022 May 20;14(5):1095. doi: 10.3390/pharmaceutics14051095. Pharmaceutics. 2022. PMID: 35631681 Free PMC article. Review.
-
Research Advances of Lipid Nanoparticles in the Treatment of Colorectal Cancer.Int J Nanomedicine. 2024 Jul 3;19:6693-6715. doi: 10.2147/IJN.S466490. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38979534 Free PMC article. Review.
-
Chemo-radiotherapy with 177Lu-PLGA(RGF)-CXCR4L for the targeted treatment of colorectal cancer.Front Med (Lausanne). 2023 Jun 12;10:1191315. doi: 10.3389/fmed.2023.1191315. eCollection 2023. Front Med (Lausanne). 2023. PMID: 37378300 Free PMC article.
References
-
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. - PubMed
-
- Mody K, Baldeo C, Bekaii-Saab T. Antiangiogenic therapy in colorectal cancer. Cancer J. 2018;24:165–170. - PubMed
LinkOut - more resources
Full Text Sources
