Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population based cohort study
- PMID: 33952445
- PMCID: PMC8097496
- DOI: 10.1136/bmj.n1114
Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: population based cohort study
Abstract
Objective: To assess rates of cardiovascular and haemostatic events in the first 28 days after vaccination with the Oxford-AstraZeneca vaccine ChAdOx1-S in Denmark and Norway and to compare them with rates observed in the general populations.
Design: Population based cohort study.
Setting: Nationwide healthcare registers in Denmark and Norway.
Participants: All people aged 18-65 years who received a first vaccination with ChAdOx1-S from 9 February 2021 to 11 March 2021. The general populations of Denmark (2016-18) and Norway (2018-19) served as comparator cohorts.
Main outcome measures: Observed 28 day rates of hospital contacts for incident arterial events, venous thromboembolism, thrombocytopenia/coagulation disorders, and bleeding among vaccinated people compared with expected rates, based on national age and sex specific background rates from the general populations of the two countries.
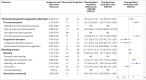
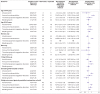
Results: The vaccinated cohorts comprised 148 792 people in Denmark (median age 45 years, 80% women) and 132 472 in Norway (median age 44 years, 78% women), who received their first dose of ChAdOx1-S. Among 281 264 people who received ChAdOx1-S, the standardised morbidity ratio for arterial events was 0.97 (95% confidence interval 0.77 to 1.20). 59 venous thromboembolic events were observed in the vaccinated cohort compared with 30 expected based on the incidence rates in the general population, corresponding to a standardised morbidity ratio of 1.97 (1.50 to 2.54) and 11 (5.6 to 17.0) excess events per 100 000 vaccinations. A higher than expected rate of cerebral venous thrombosis was observed: standardised morbidity ratio 20.25 (8.14 to 41.73); an excess of 2.5 (0.9 to 5.2) events per 100 000 vaccinations. The standardised morbidity ratio for any thrombocytopenia/coagulation disorders was 1.52 (0.97 to 2.25) and for any bleeding was 1.23 (0.97 to 1.55). 15 deaths were observed in the vaccine cohort compared with 44 expected.
Conclusions: Among recipients of ChAdOx1-S, increased rates of venous thromboembolic events, including cerebral venous thrombosis, were observed. For the remaining safety outcomes, results were largely reassuring, with slightly higher rates of thrombocytopenia/coagulation disorders and bleeding, which could be influenced by increased surveillance of vaccine recipients. The absolute risks of venous thromboembolic events were, however, small, and the findings should be interpreted in the light of the proven beneficial effects of the vaccine, the context of the given country, and the limitations to the generalisability of the study findings.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at http://www.icmje.org/coi_disclosure.pdf and declare: no support from any organisation for the submitted work. The authors report no personal conflicts of interest pertaining to this work. The Department of Clinical Epidemiology, Aarhus University Hospital, receives funding for other studies from the European Medicines Agency and companies in the form of research grants to (and administered by) Aarhus University. None of these studies have any relation to the present study. AP, JH, and LCL report participation in research projects funded by Alcon, Almirall, Astellas, AstraZeneca, Boehringer-Ingelheim, Novo Nordisk, Servier, Menarini Pharmaceuticals, and LEO Pharma, all regulator mandated phase IV studies, all with funds paid to their institution (no personal fees) and with no relation to the work reported in this paper. ØK reports participation in research projects funded by Novo Nordisk and LEO Pharma, all regulator mandated phase IV studies, all with funds paid to his institution (no personal fees) and with no relation to the work reported in this paper. HLG reports previous participation in research projects and clinical trials funded by Novo Nordisk, GSK, AstraZeneca, and Boheringer-Ingelheim, all paid to her previous institution Oslo University Hospital (no personal fees). MA has previously participated in research projects funded by Pfizer, Janssen, AstraZeneca, H Lundbeck & Mertz, and Novartis with grants received by Karolinska Institutet (no personal fees). MA has personally received fees for teaching from Atrium, the Danish Association of the Pharmaceutical Industry. The Pharmacovigilance Research Center is supported by a grant from the Novo Nordisk Foundation (NNF15SA0018404) to the University of Copenhagen.
Figures

Comment in
-
Thromboembolism and the Oxford-AstraZeneca vaccine.BMJ. 2021 May 5;373:n1159. doi: 10.1136/bmj.n1159. BMJ. 2021. PMID: 33952506 No abstract available.
References
-
- World Health Organization. COVID-19 Weekly Epidemiological Update. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situatio....
-
- Voysey M, Clemens SAC, Madhi SA, et al. Oxford COVID Vaccine Trial Group . Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021;397:99-111. 10.1016/S0140-6736(20)32661-1 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
