Vascular Senescence: A Potential Bridge Between Physiological Aging and Neurogenic Decline
- PMID: 33958987
- PMCID: PMC8093510
- DOI: 10.3389/fnins.2021.666881
Vascular Senescence: A Potential Bridge Between Physiological Aging and Neurogenic Decline
Abstract
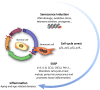
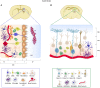
The adult mammalian brain contains distinct neurogenic niches harboring populations of neural stem cells (NSCs) with the capacity to sustain the generation of specific subtypes of neurons during the lifetime. However, their ability to produce new progeny declines with age. The microenvironment of these specialized niches provides multiple cellular and molecular signals that condition NSC behavior and potential. Among the different niche components, vasculature has gained increasing interest over the years due to its undeniable role in NSC regulation and its therapeutic potential for neurogenesis enhancement. NSCs are uniquely positioned to receive both locally secreted factors and adhesion-mediated signals derived from vascular elements. Furthermore, studies of parabiosis indicate that NSCs are also exposed to blood-borne factors, sensing and responding to the systemic circulation. Both structural and functional alterations occur in vasculature with age at the cellular level that can affect the proper extrinsic regulation of NSCs. Additionally, blood exchange experiments in heterochronic parabionts have revealed that age-associated changes in blood composition also contribute to adult neurogenesis impairment in the elderly. Although the mechanisms of vascular- or blood-derived signaling in aging are still not fully understood, a general feature of organismal aging is the accumulation of senescent cells, which act as sources of inflammatory and other detrimental signals that can negatively impact on neighboring cells. This review focuses on the interactions between vascular senescence, circulating pro-senescence factors and the decrease in NSC potential during aging. Understanding the mechanisms of NSC dynamics in the aging brain could lead to new therapeutic approaches, potentially include senolysis, to target age-dependent brain decline.
Keywords: adult neural stem cell; endothelial cell senescence; neurogenic niche; parabiosis; senescence-associated secretory phenotype.
Copyright © 2021 Rojas-Vázquez, Blasco-Chamarro, López-Fabuel, Martínez-Máñez and Fariñas.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Grafted Subventricular Zone Neural Stem Cells Display Robust Engraftment and Similar Differentiation Properties and Form New Neurogenic Niches in the Young and Aged Hippocampus.Stem Cells Transl Med. 2016 Sep;5(9):1204-15. doi: 10.5966/sctm.2015-0270. Epub 2016 May 18. Stem Cells Transl Med. 2016. PMID: 27194744 Free PMC article.
-
Exosomes as Novel Regulators of Adult Neurogenic Niches.Front Cell Neurosci. 2016 Jan 19;9:501. doi: 10.3389/fncel.2015.00501. eCollection 2015. Front Cell Neurosci. 2016. PMID: 26834560 Free PMC article. Review.
-
Role of Astrocytes in the Neurogenic Niches.Methods Mol Biol. 2019;1938:19-33. doi: 10.1007/978-1-4939-9068-9_2. Methods Mol Biol. 2019. PMID: 30617970 Review.
-
Heterochronic parabiosis regulates the extent of cellular senescence in multiple tissues.Geroscience. 2020 Jun;42(3):951-961. doi: 10.1007/s11357-020-00185-1. Epub 2020 Apr 13. Geroscience. 2020. PMID: 32285290 Free PMC article.
-
The systemic environment: at the interface of aging and adult neurogenesis.Cell Tissue Res. 2018 Jan;371(1):105-113. doi: 10.1007/s00441-017-2715-8. Epub 2017 Nov 9. Cell Tissue Res. 2018. PMID: 29124393 Free PMC article. Review.
Cited by
-
A renal clearable fluorogenic probe for in vivo β-galactosidase activity detection during aging and senolysis.Nat Commun. 2024 Jan 26;15(1):775. doi: 10.1038/s41467-024-44903-1. Nat Commun. 2024. PMID: 38278798 Free PMC article.
-
Among Gerontogens, Heavy Metals Are a Class of Their Own: A Review of the Evidence for Cellular Senescence.Brain Sci. 2023 Mar 16;13(3):500. doi: 10.3390/brainsci13030500. Brain Sci. 2023. PMID: 36979310 Free PMC article. Review.
-
Old blood from heterochronic parabionts accelerates vascular aging in young mice: transcriptomic signature of pathologic smooth muscle remodeling.Geroscience. 2022 Apr;44(2):953-981. doi: 10.1007/s11357-022-00519-1. Epub 2022 Feb 5. Geroscience. 2022. PMID: 35124764 Free PMC article.
-
The Brain and Spinal Microvasculature in Normal Aging.J Gerontol A Biol Sci Med Sci. 2023 Aug 2;78(8):1309-1319. doi: 10.1093/gerona/glad107. J Gerontol A Biol Sci Med Sci. 2023. PMID: 37093786 Free PMC article. Review.
-
The vascular microenvironment and its stem cells regulate vascular homeostasis.Front Cell Dev Biol. 2025 Mar 6;13:1544129. doi: 10.3389/fcell.2025.1544129. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40114970 Free PMC article. Review.
References
-
- Apostolopoulou M., Kiehl T. R., Winter M., Cardenas De La Hoz E., Boles N. C., Bjornsson C. S., et al. (2017). Non-monotonic changes in progenitor cell behavior and gene expression during aging of the adult V-SVZ neural stem cell niche. Stem Cell Rep. 9 1931–1947. 10.1016/j.stemcr.2017.10.005 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

