Artificially altered gravity elicits cell homeostasis imbalance in planarian worms, and cerium oxide nanoparticles counteract this effect
- PMID: 33960131
- PMCID: PMC8518838
- DOI: 10.1002/jbm.a.37215
Artificially altered gravity elicits cell homeostasis imbalance in planarian worms, and cerium oxide nanoparticles counteract this effect
Abstract
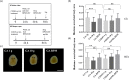
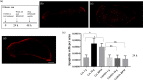
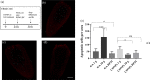
Gravity alterations elicit complex and mostly detrimental effects on biological systems. Among these, a prominent role is occupied by oxidative stress, with consequences for tissue homeostasis and development. Studies in altered gravity are relevant for both Earth and space biomedicine, but their implementation using whole organisms is often troublesome. Here we utilize planarians, simple worm model for stem cell and regeneration biology, to characterize the pathogenic mechanisms brought by artificial gravity alterations. In particular, we provide a comprehensive evaluation of molecular responses in intact and regenerating specimens, and demonstrate a protective action from the space-apt for nanotechnological antioxidant cerium oxide nanoparticles.
Keywords: cerium oxide nanoparticles; hypergravity; microgravity; planarians; stem cells.
© 2021 The Authors. Journal of Biomedical Materials Research Part A published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare no potential conflict of interest.
Figures





Similar articles
-
The first inorganic mitogens: Cerium oxide and cerium fluoride nanoparticles stimulate planarian regeneration via neoblastic activation.Mater Sci Eng C Mater Biol Appl. 2019 Nov;104:109924. doi: 10.1016/j.msec.2019.109924. Epub 2019 Jun 27. Mater Sci Eng C Mater Biol Appl. 2019. PMID: 31499991
-
Stem cell and tissue regeneration analysis in low-dose irradiated planarians treated with cerium oxide nanoparticles.Mater Sci Eng C Mater Biol Appl. 2020 Oct;115:111113. doi: 10.1016/j.msec.2020.111113. Epub 2020 May 25. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 32600713
-
Toxicity of ceria nanoparticles to the regeneration of freshwater planarian Dugesia japonica: The role of biotransformation.Sci Total Environ. 2023 Jan 20;857(Pt 3):159590. doi: 10.1016/j.scitotenv.2022.159590. Epub 2022 Oct 19. Sci Total Environ. 2023. PMID: 36270358
-
Exploring the innovative application of cerium oxide nanoparticles for addressing oxidative stress in ovarian tissue regeneration.J Ovarian Res. 2024 Dec 5;17(1):241. doi: 10.1186/s13048-024-01566-2. J Ovarian Res. 2024. PMID: 39633503 Free PMC article. Review.
-
Protective potential of cerium oxide nanoparticles in diabetes mellitus.J Trace Elem Med Biol. 2021 Jul;66:126742. doi: 10.1016/j.jtemb.2021.126742. Epub 2021 Mar 10. J Trace Elem Med Biol. 2021. PMID: 33773280 Review.
Cited by
-
Cell quiescence in planarian stem cells, interplay between p53 and nutritional stimuli.Open Biol. 2022 Dec;12(12):220216. doi: 10.1098/rsob.220216. Epub 2022 Dec 21. Open Biol. 2022. PMID: 36541101 Free PMC article.
References
-
- Brown AH. The influence of gravity on the structure and functions of plant material. Adv Space Res. 1984;4(12):299‐303. - PubMed
-
- Gualandris‐Parisot L, Husson D, Foulquier F, et al. Pleurodeles waltl, amphibian, Urodele, is a suitable biological model for embryological and physiological space experiments on a vertebrate. Adv Space Res. 2001;28(4):569‐578. - PubMed
-
- Grigoryan EN, Dvorochkin N, Poplinskaya VA, Yousuf R, Radugina EA, Almeida EA. The effect of hypergravity on the lens, cornea and tail regeneration in Urodela. Acta Astronaut. 2017;138:423‐433.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

