Adipocytes Provide Fatty Acids to Acute Lymphoblastic Leukemia Cells
- PMID: 33968771
- PMCID: PMC8100891
- DOI: 10.3389/fonc.2021.665763
Adipocytes Provide Fatty Acids to Acute Lymphoblastic Leukemia Cells
Abstract
Background: There is increasing evidence that adipocytes play an active role in the cancer microenvironment. We have previously reported that adipocytes interact with acute lymphoblastic leukemia (ALL) cells, contributing to chemotherapy resistance and treatment failure. In the present study, we investigated whether part of this resistance is due to adipocyte provision of lipids to ALL cells.
Methods: We cultured 3T3-L1 adipocytes, and tested whether ALL cells or ALL-released cytokines induced FFA release. We investigated whether ALL cells took up these FFA, and using fluorescent tagged BODIPY-FFA and lipidomics, evaluated which lipid moieties were being transferred from adipocytes to ALL. We evaluated the effects of adipocyte-derived lipids on ALL cell metabolism using a Seahorse XF analyzer and expression of enzymes important for lipid metabolism, and tested whether these lipids could protect ALL cells from chemotherapy. Finally, we evaluated a panel of lipid synthesis and metabolism inhibitors to determine which were affected by the presence of adipocytes.
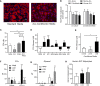
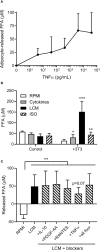
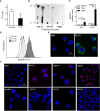
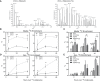
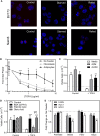
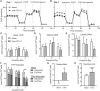
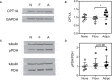
Results: Adipocytes release free fatty acids (FFA) when in the presence of ALL cells. These FFA are taken up by the ALL cells and incorporated into triglycerides and phospholipids. Some of these lipids are stored in lipid droplets, which can be utilized in states of fuel deprivation. Adipocytes preferentially release monounsaturated FFA, and this can be attenuated by inhibiting the desaturating enzyme steroyl-CoA decarboxylase-1 (SCD1). Adipocyte-derived FFA can relieve ALL cell endogenous lipogenesis and reverse the cytotoxicity of pharmacological acetyl-CoA carboxylase (ACC) inhibition. Further, adipocytes alter ALL cell metabolism, shifting them from glucose to FFA oxidation. Interestingly, the unsaturated fatty acid, oleic acid, protects ALL cells from modest concentrations of chemotherapy, such as those that might be present in the ALL microenvironment. In addition, targeting lipid synthesis and metabolism can potentially reverse adipocyte protection of ALL cells.
Conclusion: These findings uncover a previously unidentified interaction between ALL cells and adipocytes, leading to transfer of FFA for use as a metabolic fuel and macromolecule building block. This interaction may contribute to ALL resistance to chemotherapy, and could potentially be targeted to improve ALL treatment outcome.
Keywords: FFA; adipocytes; leukemia; lipid droplets; microenvironment.
Copyright © 2021 Tucci, Chen, Margulis, Orgel, Paszkiewicz, Cohen, Oberley, Wahhab, Jones, Divakaruni, Hsu, Noll, Sheng, Zare and Mittelman.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Ethanol extracts of chickpeas alter the total lipid content and expression levels of genes related to fatty acid metabolism in mouse 3T3-L1 adipocytes.Int J Mol Med. 2016 Aug;38(2):574-84. doi: 10.3892/ijmm.2016.2654. Epub 2016 Jun 24. Int J Mol Med. 2016. PMID: 27353085 Free PMC article.
-
FFA-induced adipocyte inflammation and insulin resistance: involvement of ER stress and IKKβ pathways.Obesity (Silver Spring). 2011 Mar;19(3):483-91. doi: 10.1038/oby.2010.200. Epub 2010 Sep 9. Obesity (Silver Spring). 2011. PMID: 20829802
-
Regulation of acetyl CoA carboxylase and carnitine palmitoyl transferase-1 in rat adipocytes.Obes Res. 2005 Sep;13(9):1530-9. doi: 10.1038/oby.2005.188. Obes Res. 2005. PMID: 16222055
-
Metabolic Remodeling Induced by Adipocytes: A New Achilles' Heel in Invasive Breast Cancer?Curr Med Chem. 2020;27(24):3984-4001. doi: 10.2174/0929867325666180426165001. Curr Med Chem. 2020. PMID: 29708068 Review.
-
Fat Cell and Fatty Acid Turnover in Obesity.Adv Exp Med Biol. 2017;960:135-160. doi: 10.1007/978-3-319-48382-5_6. Adv Exp Med Biol. 2017. PMID: 28585198 Review.
Cited by
-
Autophagy and Metabolism in Normal and Malignant Hematopoiesis.Int J Mol Sci. 2021 Aug 9;22(16):8540. doi: 10.3390/ijms22168540. Int J Mol Sci. 2021. PMID: 34445246 Free PMC article. Review.
-
Molecular Deconvolution of Bone Marrow Adipose Tissue Interactions with Malignant Hematopoiesis: Potential for New Therapy Development.Curr Osteoporos Rep. 2024 Aug;22(4):367-377. doi: 10.1007/s11914-024-00879-x. Epub 2024 Jun 26. Curr Osteoporos Rep. 2024. PMID: 38922359 Review.
-
Leveraging altered lipid metabolism in treating B cell malignancies.Prog Lipid Res. 2024 Jul;95:101288. doi: 10.1016/j.plipres.2024.101288. Epub 2024 Jul 2. Prog Lipid Res. 2024. PMID: 38964473 Free PMC article. Review.
-
The regulation, function, and role of lipophagy, a form of selective autophagy, in metabolic disorders.Cell Death Dis. 2022 Feb 8;13(2):132. doi: 10.1038/s41419-022-04593-3. Cell Death Dis. 2022. PMID: 35136038 Free PMC article. Review.
-
Body mass trajectory from diagnosis to the end of treatment in a pediatric acute lymphoblastic leukemia cohort.Sci Rep. 2023 Aug 21;13(1):13590. doi: 10.1038/s41598-023-39287-z. Sci Rep. 2023. PMID: 37604919 Free PMC article.
References
-
- Surveillance,Epidemiology. End Results (SEER) Program . Seer*Stat Database (2018). Available at: www.seer.cancer.gov.

