Antibodies and Vaccines Target RBD of SARS-CoV-2
- PMID: 33968996
- PMCID: PMC8100443
- DOI: 10.3389/fmolb.2021.671633
Antibodies and Vaccines Target RBD of SARS-CoV-2
Abstract
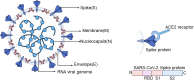
The novel human coronavirus, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), which gives rise to the coronavirus disease 2019 (COVID-19), has caused a serious threat to global public health. On March 11, 2020, the WHO had officially announced COVID-19 as a pandemic. Therefore, it is vital to find effective and safe neutralizing antibodies and vaccines for COVID-19. The critical neutralizing domain (CND) that is contained in the receptor-binding domain (RBD) of the spike protein (S protein) could lead to a highly potent neutralizing antibody response as well as the cross-protection of other strains of SARS. By using RBD as an antigen, many neutralizing antibodies are isolated that are essential to the therapeutics of COVID-19. Furthermore, a subunit vaccine, which is based on the RBD, is expected to be safer than others, thus the RBD in the S protein is a more important target for vaccine development. In this review, we focus on neutralizing antibodies that are targeting RBD as well as the vaccine based on RBD under current development.
Keywords: COVID-19; RBD; SARS-CoV-2; antibodies; vaccines.
Copyright © 2021 Min and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Structural Basis of a Human Neutralizing Antibody Specific to the SARS-CoV-2 Spike Protein Receptor-Binding Domain.Microbiol Spectr. 2021 Oct 31;9(2):e0135221. doi: 10.1128/Spectrum.01352-21. Epub 2021 Oct 13. Microbiol Spectr. 2021. PMID: 34643438 Free PMC article.
-
Receptor-binding domain as a target for developing SARS vaccines.J Thorac Dis. 2013 Aug;5 Suppl 2(Suppl 2):S142-8. doi: 10.3978/j.issn.2072-1439.2013.06.06. J Thorac Dis. 2013. PMID: 23977435 Free PMC article.
-
A Glycosylated RBD Protein Induces Enhanced Neutralizing Antibodies against Omicron and Other Variants with Improved Protection against SARS-CoV-2 Infection.J Virol. 2022 Sep 14;96(17):e0011822. doi: 10.1128/jvi.00118-22. Epub 2022 Aug 16. J Virol. 2022. PMID: 35972290 Free PMC article.
-
Targeting SARS-CoV2 Spike Protein Receptor Binding Domain by Therapeutic Antibodies.Biomed Pharmacother. 2020 Oct;130:110559. doi: 10.1016/j.biopha.2020.110559. Epub 2020 Aug 1. Biomed Pharmacother. 2020. PMID: 32768882 Free PMC article. Review.
-
SARS-CoV-2 Spike Protein Extrapolation for COVID Diagnosis and Vaccine Development.Front Mol Biosci. 2021 Jul 28;8:607886. doi: 10.3389/fmolb.2021.607886. eCollection 2021. Front Mol Biosci. 2021. PMID: 34395515 Free PMC article. Review.
Cited by
-
L-shaped distribution of the relative substitution rate (c/μ) observed for SARS-COV-2's genome, inconsistent with the selectionist theory, the neutral theory and the nearly neutral theory but a near-neutral balanced selection theory: Implication on "neutralist-selectionist" debate.Comput Biol Med. 2023 Feb;153:106522. doi: 10.1016/j.compbiomed.2022.106522. Epub 2023 Jan 5. Comput Biol Med. 2023. PMID: 36638615 Free PMC article.
-
A Review: Understanding Molecular Mechanisms of Antibody-Dependent Enhancement in Viral Infections.Vaccines (Basel). 2023 Jul 14;11(7):1240. doi: 10.3390/vaccines11071240. Vaccines (Basel). 2023. PMID: 37515055 Free PMC article. Review.
-
SARS-CoV-2 spike protein reduces burst activities in neurons measured by micro-electrode arrays.Ann Med Surg (Lond). 2023 Jun 10;85(7):3469-3476. doi: 10.1097/MS9.0000000000000950. eCollection 2023 Jul. Ann Med Surg (Lond). 2023. PMID: 37427167 Free PMC article.
-
Review of therapeutic mechanisms and applications based on SARS-CoV-2 neutralizing antibodies.Front Microbiol. 2023 Mar 16;14:1122868. doi: 10.3389/fmicb.2023.1122868. eCollection 2023. Front Microbiol. 2023. PMID: 37007494 Free PMC article. Review.
-
Co-immunization with spike and nucleocapsid based DNA vaccines for long-term protective immunity against SARS-CoV-2 Omicron.NPJ Vaccines. 2024 Dec 19;9(1):252. doi: 10.1038/s41541-024-01043-3. NPJ Vaccines. 2024. PMID: 39702529 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

