The Intersection of COVID-19 and Autoimmunity: What is Our Current Understanding?
- PMID: 33969248
- PMCID: PMC8097827
- DOI: 10.20411/pai.v6i1.417
The Intersection of COVID-19 and Autoimmunity: What is Our Current Understanding?
Abstract
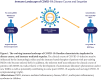
Viral infections have historically had a complex relationship with autoimmune diseases. For patients with preexisting autoimmune disorders, often complicated by immunosuppressive therapies, there are numerous potential effects of COVID-19, a disease of complex immunobiology, including the potential for an altered natural history of COVID-19 when infected. In addition, individuals without recognized autoimmune disease may be vulnerable to virally induced autoimmunity in the forms of autoantibody formation, as well as the development of clinical immune-mediated inflammatory diseases. Until quite recently in the pandemic, this relationship between COVID-19 and autoimmune diseases has been relatively underexplored; yet such investigation offers potential insights into immunopathogenesis as well as for the development of new immune-based therapeutics. Our review examines this relationship through exploration of a series of questions with relevance to both immunopathogenic mechanisms as well as some clinical implications.
Keywords: Autoantibodies; Autoimmunity; COVID-19; DMARDs; Immune-mediated inflammatory disease; MIS-C; SARS-CoV-2.
Copyright © Pathogens and Immunity 2021.
Conflict of interest statement
Consulting fees from AbbVie, BMS, Crescendo, Genentech, Gilead, GSK, Horizon, Janssen, Novartis, and Sanofi; speaking fees from AbbVie, Crescendo, Genentech, Janssen, Novartis, and Sanofi.
Figures

References
-
- Murphy K, Weaver C.. Janeway's Immunobiology. New York: Garland Science; 2017. 729-44.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
