Circuit organization of the rodent medial prefrontal cortex
- PMID: 33972100
- PMCID: PMC8222144
- DOI: 10.1016/j.tins.2021.03.006
Circuit organization of the rodent medial prefrontal cortex
Abstract
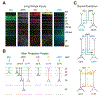
The prefrontal cortex (PFC) orchestrates higher brain function and becomes disrupted in many mental health disorders. The rodent medial PFC (mPFC) possesses an enormous variety of projection neurons and interneurons. These cells are engaged by long-range inputs from other brain regions involved in cognition, motivation, and emotion. They also communicate in the local network via specific connections between excitatory and inhibitory cells. In this review, we describe the cellular diversity of the rodent mPFC, the impact of long-range afferents, and the specificity of local microcircuits. We highlight similarities with and differences between other cortical areas, illustrating how the circuit organization of the mPFC may give rise to its unique functional roles.
Keywords: cell types; circuits; interneuron; prefrontal cortex; projection neuron; synapses.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no interests in relation to this work.
Figures
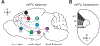

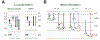


References
-
- Miller EK and Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24, 167–202 - PubMed
-
- Arnsten AFT and Rubia K (2012) Neurobiological Circuits Regulating Attention, Cognitive Control, Motivation, and Emotion: Disruptions in Neurodevelopmental Psychiatric Disorders. Journal of the American Academy of Child & Adolescent Psychiatry 51, 356–367 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

