Recent advances in the chemistry of ketyl radicals
- PMID: 33972956
- PMCID: PMC8111543
- DOI: 10.1039/d0cs00358a
Recent advances in the chemistry of ketyl radicals
Abstract
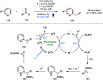
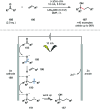
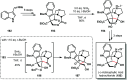
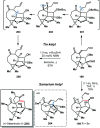
Ketyl radicals are valuable reactive intermediates for synthesis and are used extensively to construct complex, functionalized products from carbonyl substrates. Single electron transfer (SET) reduction of the C[double bond, length as m-dash]O bond of aldehydes and ketones is the classical approach for the formation of ketyl radicals and metal reductants are the archetypal reagents employed. The past decade has, however, witnessed significant advances in the generation and harnessing of ketyl radicals. This tutorial review highlights recent, exciting developments in the chemistry of ketyl radicals by comparing the varied contemporary - for example, using photoredox catalysts - and more classical approaches for the generation and use of ketyl radicals. The review will focus on different strategies for ketyl radical generation, their creative use in new synthetic protocols, strategies for the control of enantioselectivity, and detailed mechanisms where appropriate.
Conflict of interest statement
There are no conflicts to declare.
Figures


















Similar articles
-
Recent developments in transition-metal photoredox-catalysed reactions of carbonyl derivatives.Chem Commun (Camb). 2017 Dec 7;53(98):13093-13112. doi: 10.1039/c7cc06287g. Chem Commun (Camb). 2017. PMID: 29125152 Free PMC article.
-
When Light Meets Nitrogen-Centered Radicals: From Reagents to Catalysts.Acc Chem Res. 2020 May 19;53(5):1066-1083. doi: 10.1021/acs.accounts.0c00090. Epub 2020 Apr 14. Acc Chem Res. 2020. PMID: 32286794
-
Synthetic Utilization of α-Aminoalkyl Radicals and Related Species in Visible Light Photoredox Catalysis.Acc Chem Res. 2016 Sep 20;49(9):1946-56. doi: 10.1021/acs.accounts.6b00251. Epub 2016 Aug 9. Acc Chem Res. 2016. PMID: 27505299
-
Visible-Light Photocatalysis of the Ketyl Radical Coupling Reaction.Chemistry. 2019 Feb 26;25(12):2949-2961. doi: 10.1002/chem.201804873. Epub 2018 Dec 14. Chemistry. 2019. PMID: 30414339 Review.
-
Recent application of visible-light induced radicals in C-S bond formation.RSC Adv. 2020 May 27;10(34):20046-20056. doi: 10.1039/d0ra03086d. eCollection 2020 May 26. RSC Adv. 2020. Retraction in: RSC Adv. 2024 Sep 5;14(39):28345. doi: 10.1039/d4ra90096k. PMID: 35520400 Free PMC article. Retracted. Review.
Cited by
-
Synthesis of alcohols: streamlined C1 to Cn hydroxyalkylation through photoredox catalysis.Chem Sci. 2024 Jun 13;15(29):11337-11346. doi: 10.1039/d4sc02696a. eCollection 2024 Jul 24. Chem Sci. 2024. PMID: 39055000 Free PMC article.
-
Direct photo-induced reductive Heck cyclization of indoles for the efficient preparation of polycyclic indolinyl compounds.Chem Sci. 2021 Sep 16;12(42):14050-14058. doi: 10.1039/d1sc04258k. eCollection 2021 Nov 3. Chem Sci. 2021. PMID: 34760188 Free PMC article.
-
Sustainable radical approaches for cross electrophile coupling to synthesize trifluoromethyl- and allyl-substituted tert-alcohols.iScience. 2021 Oct 30;24(12):103388. doi: 10.1016/j.isci.2021.103388. eCollection 2021 Dec 17. iScience. 2021. PMID: 34841228 Free PMC article.
-
A Persistent Phosphanyl-Substituted Thioketyl Radical Anion.Angew Chem Int Ed Engl. 2022 Feb 21;61(9):e202114792. doi: 10.1002/anie.202114792. Epub 2022 Jan 12. Angew Chem Int Ed Engl. 2022. PMID: 34843637 Free PMC article.
-
Decatungstate-catalyzed radical disulfuration through direct C-H functionalization for the preparation of unsymmetrical disulfides.Nat Commun. 2022 Jul 6;13(1):3886. doi: 10.1038/s41467-022-31617-5. Nat Commun. 2022. PMID: 35794128 Free PMC article.
References
-
- Kratish Y. Pinchuk D. Kaushansky A. Molev V. Tumanskii B. Bravo-Zhivotovskii D. Apeloig Y. Angew. Chem., Int. Ed. 2019;58:18849–18853. - PubMed
-
and references therein
-
- Nocera G. Young A. Palumbo F. Emery K. J. Coulthard G. McGuire T. Tuttle T. Murphy J. A. J. Am. Chem. Soc. 2018;140:9751–9757. - PubMed
-
- Yamamoto Y. Hattori R. Miwa T. Nakagai Y.-I. Kubota T. Yamamoto C. Okamoto Y. Itoh K. J. Org. Chem. 2001;66:3865–3870. - PubMed
-
- Fürstner A. Shi N. J. Am. Chem. Soc. 1996;118:2533–2534.
-
- Bichovski P. Haas T. M. Kratzert D. Streuff J. Chem. – Eur. J. 2015;21:2339–2342. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

