Long-term outcomes after gene therapy for adenosine deaminase severe combined immune deficiency
- PMID: 33974038
- PMCID: PMC8525336
- DOI: 10.1182/blood.2020010260
Long-term outcomes after gene therapy for adenosine deaminase severe combined immune deficiency
Abstract
Patients lacking functional adenosine deaminase activity have severe combined immunodeficiency (ADA SCID), which can be treated with ADA enzyme replacement therapy (ERT), allogeneic hematopoietic stem cell transplantation (HSCT), or autologous HSCT with gene-corrected cells (gene therapy [GT]). A cohort of 10 ADA SCID patients, aged 3 months to 15 years, underwent GT in a phase 2 clinical trial between 2009 and 2012. Autologous bone marrow CD34+ cells were transduced ex vivo with the MND (myeloproliferative sarcoma virus, negative control region deleted, dl587rev primer binding site)-ADA gammaretroviral vector (gRV) and infused following busulfan reduced-intensity conditioning. These patients were monitored in a long-term follow-up protocol over 8 to 11 years. Nine of 10 patients have sufficient immune reconstitution to protect against serious infections and have not needed to resume ERT or proceed to secondary allogeneic HSCT. ERT was restarted 6 months after GT in the oldest patient who had no evidence of benefit from GT. Four of 9 evaluable patients with the highest gene marking and B-cell numbers remain off immunoglobulin replacement therapy and responded to vaccines. There were broad ranges of responses in normalization of ADA enzyme activity and adenine metabolites in blood cells and levels of cellular and humoral immune reconstitution. Outcomes were generally better in younger patients and those receiving higher doses of gene-marked CD34+ cells. No patient experienced a leukoproliferative event after GT, despite persisting prominent clones with vector integrations adjacent to proto-oncogenes. These long-term findings demonstrate enduring efficacy of GT for ADA SCID but also highlight risks of genotoxicity with gRVs. This trial was registered at www.clinicaltrials.gov as #NCT00794508.
© 2021 by The American Society of Hematology.
Figures

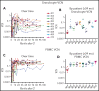
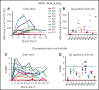
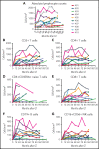


Comment in
-
Built to last: gene therapy for ADA SCID.Blood. 2021 Oct 14;138(15):1287-1288. doi: 10.1182/blood.2021012300. Blood. 2021. PMID: 34647983 Free PMC article. No abstract available.
References
-
- Giblett ER, Anderson JE, Cohen F, Pollara B, Meuwissen HJ.. Adenosine-deaminase deficiency in two patients with severely impaired cellular immunity. Lancet. 1972;2(7786):1067-1069. - PubMed
-
- Hershfield M, Adam MP, Ardinger HH, et al. Adenosine deaminase deficiency. pubmed.ncbi.nlm.nih.gov/20301656/. Accessed August 30, 2020.
-
- Blackburn MR, Kellems RE.. Adenosine deaminase deficiency: metabolic basis of immune deficiency and pulmonary inflammation. Adv Immunol. 2005; 86(1):41. - PubMed
-
- Parkman R, Gelfand EW, Rosen FS, Sanderson A, Hirschhorn R.. Severe combined immunodeficiency and adenosine deaminase deficiency. N Engl J Med. 1975;292(14):714-719. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

