Treatment timing and the effects of rhythm control strategy in patients with atrial fibrillation: nationwide cohort study
- PMID: 33975876
- PMCID: PMC8111568
- DOI: 10.1136/bmj.n991
Treatment timing and the effects of rhythm control strategy in patients with atrial fibrillation: nationwide cohort study
Abstract
Objective: To investigate whether the results of a rhythm control strategy differ according to the duration between diagnosis of atrial fibrillation and treatment initiation.
Design: Longitudinal observational cohort study.
Setting: Population based cohort from the Korean National Health Insurance Service database.
Participants: 22 635 adults with atrial fibrillation and cardiovascular conditions, newly treated with rhythm control (antiarrhythmic drugs or ablation) or rate control strategies between 28 July 2011 and 31 December 2015.
Main outcome measure: A composite outcome of death from cardiovascular causes, ischaemic stroke, admission to hospital for heart failure, or acute myocardial infarction.
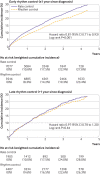
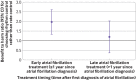
Results: Of the study population, 12 200 (53.9%) were male, the median age was 70, and the median follow-up duration was 2.1 years. Among patients with early treatment for atrial fibrillation (initiated within one year since diagnosis), compared with rate control, rhythm control was associated with a lower risk of the primary composite outcome (weighted incidence rate per 100 person years 7.42 in rhythm control v 9.25 in rate control; hazard ratio 0.81, 95% confidence interval 0.71 to 0.93; P=0.002). No difference in the risk of the primary composite outcome was found between rhythm and rate control (weighted incidence rate per 100 person years 8.67 in rhythm control v 8.99 in rate control; 0.97, 0.78 to 1.20; P=0.76) in patients with late treatment for atrial fibrillation (initiated after one year since diagnosis). No significant differences in safety outcomes were found between the rhythm and rate control strategies across different treatment timings. Earlier initiation of treatment was linearly associated with more favourable cardiovascular outcomes for rhythm control compared with rate control.
Conclusions: Early initiation of rhythm control treatment was associated with a lower risk of adverse cardiovascular outcomes than rate control treatment in patients with recently diagnosed atrial fibrillation. This association was not found in patients who had had atrial fibrillation for more than one year.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; GYHL has served as a consultant for Bayer/Janssen, BMS/Pfizer, Biotronik, Medtronic, Boehringer Ingelheim, Novartis, Verseon, and Daiichi-Sankyo and as a speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim, and Daiichi-Sankyo. No fees have been received directly or personally. BJ has served as a speaker for Bayer, BMS/Pfizer, Medtronic, and Daiichi-Sankyo and received research funds from Medtronic and Abbott. No fees have been received directly or personally. The remaining authors have no other relationships or activities that could appear to have influenced the submitted work.
Figures





References
-
- Hindricks G, Potpara T, Dagres N, et al. ESC Scientific Document Group . 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2021;42:373-498. 10.1093/eurheartj/ehaa612 - DOI - PubMed
-
- Marijon E, Le Heuzey JY, Connolly S, et al. RE-LY Investigators . Causes of death and influencing factors in patients with atrial fibrillation: a competing-risk analysis from the randomized evaluation of long-term anticoagulant therapy study. Circulation 2013;128:2192-201. 10.1161/CIRCULATIONAHA.112.000491 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
