Alternative reproductive tactics in male freshwater fish influence the accuracy of species recognition
- PMID: 33976782
- PMCID: PMC8093699
- DOI: 10.1002/ece3.7267
Alternative reproductive tactics in male freshwater fish influence the accuracy of species recognition
Abstract
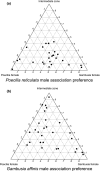
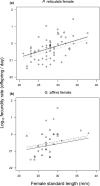
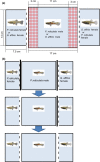
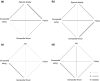
Sexual conflict can result in coercive mating. Because males bear low costs of heterospecific mating, coercive males may engage in misdirected mating attempts toward heterospecific females. In contrast, sexual selection through consensual mate choice can cause mate recognition cues among species to diverge, leading to more accurate species recognition. Some species show both coercive mating and mate choice-associated courtship behaviors as male alternative reproductive tactics. We hypothesized that if the selection pressures on each tactic differ, then the accuracy of species recognition would also change depending on the mating tactic adopted. We tested this hypothesis in the guppy (Poecilia reticulata) and mosquitofish (Gambusia affinis) by a series of choice experiments. Poecilia reticulata and G. affinis males both showed imperfect species recognition and directed all components of mating behavior toward heterospecific females. They tended to direct courtship displays more frequently toward conspecific than heterospecific females. With male P. reticulata, however, accurate species recognition disappeared when they attempted coercive copulation: they directed coercions more frequently toward heterospecific females. We also found that heterospecific sexual interaction had little effect on the fecundity of gravid females, which suggests that prepregnancy interactions likely underpin the exclusion of G. affinis by P. reticulata in our region.
Keywords: Fisherian process; Poeciliidae; male mate choice; mate recognition; reproductive interference.
© 2021 The Authors. Ecology and Evolution published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Plasticity of the mate choice mind: courtship evokes choice-like brain responses in females from a coercive mating system.Genes Brain Behav. 2014 Apr;13(4):365-75. doi: 10.1111/gbb.12124. Epub 2014 Mar 19. Genes Brain Behav. 2014. PMID: 24548673
-
Sexual Selection in Mosquitofish: Differences in the Use of Mating Cues Between Sexes.Animals (Basel). 2025 May 21;15(10):1489. doi: 10.3390/ani15101489. Animals (Basel). 2025. PMID: 40427366 Free PMC article.
-
Effects of a parasitic nematode on male mate choice in a livebearing fish with a coercive mating system (western mosquitofish, Gambusia affinis).Behav Processes. 2009 Jan;80(1):1-6. doi: 10.1016/j.beproc.2008.07.010. Epub 2008 Aug 14. Behav Processes. 2009. PMID: 18765273
-
Male size and reproductive performance in three species of livebearing fishes (Gambusia spp.): A systematic review and meta-analysis.J Anim Ecol. 2021 Oct;90(10):2431-2445. doi: 10.1111/1365-2656.13554. Epub 2021 Jul 6. J Anim Ecol. 2021. PMID: 34231219
-
The evolution of male mate choice in insects: a synthesis of ideas and evidence.Biol Rev Camb Philos Soc. 2001 Aug;76(3):305-39. doi: 10.1017/s1464793101005693. Biol Rev Camb Philos Soc. 2001. PMID: 11569787 Review.
Cited by
-
Hybridization in the Anthropocene - how pollution and climate change disrupt mate selection in freshwater fish.Biol Rev Camb Philos Soc. 2025 Feb;100(1):35-49. doi: 10.1111/brv.13126. Epub 2024 Aug 2. Biol Rev Camb Philos Soc. 2025. PMID: 39092475 Free PMC article.
-
Heterospecific interaction in two beetle species: Males with weapons decrease the reproductive success of species with weaponless males.Ecol Evol. 2024 Jun 18;14(6):e11518. doi: 10.1002/ece3.11518. eCollection 2024 Jun. Ecol Evol. 2024. PMID: 38895581 Free PMC article.
References
-
- Arnqvist, G. , & Rowe, L. (2005). Sexual conflict. Princeton University Press.
-
- Chung, H. , Loehlin, D. W. , Dufour, H. D. , Vacarro, K. , Millar, J. G. , & Carroll, S. B. (2014). A single gene affects both ecological divergence and mate choice in Drosophila. Science, 343(6175), 1148–1151. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources

