Rapid development and deployment of high-volume vaccines for pandemic response
- PMID: 33977274
- PMCID: PMC7361221
- DOI: 10.1002/amp2.10060
Rapid development and deployment of high-volume vaccines for pandemic response
Abstract
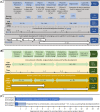
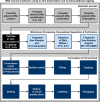
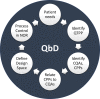
Overcoming pandemics, such as the current Covid-19 outbreak, requires the manufacture of several billion doses of vaccines within months. This is an extremely challenging task given the constraints in small-scale manufacturing for clinical trials, clinical testing timelines involving multiple phases and large-scale drug substance and drug product manufacturing. To tackle these challenges, regulatory processes are fast-tracked, and rapid-response manufacturing platform technologies are used. Here, we evaluate the current progress, challenges ahead and potential solutions for providing vaccines for pandemic response at an unprecedented scale and rate. Emerging rapid-response vaccine platform technologies, especially RNA platforms, offer a high productivity estimated at over 1 billion doses per year with a small manufacturing footprint and low capital cost facilities. The self-amplifying RNA (saRNA) drug product cost is estimated at below 1 USD/dose. These manufacturing processes and facilities can be decentralized to facilitate production, distribution, but also raw material supply. The RNA platform technology can be complemented by an a priori Quality by Design analysis aided by computational modeling in order to assure product quality and further speed up the regulatory approval processes when these platforms are used for epidemic or pandemic response in the future.
Keywords: Quality by Design; RNA vaccines; bioprocess modeling; distributed manufacturing; pandemic‐response vaccine manufacturing; techno‐economic modeling; vaccine platform technology.
© 2020 The Authors. Journal of Advanced Manufacturing and Processing published by Wiley Periodicals LLC. on behalf of American Institute of Chemical Engineers.
Figures
References
-
- Rémy V., Zöllner Y., Heckmann U., J. Market Access Health Policy 2015, 3, 1. 10.3402/jmahp.v3.27041, https://pubmed.ncbi.nlm.nih.gov/27123189. - DOI - PMC - PubMed
-
- Remy V., Largeron N., Quilici S., Carroll S., J. Market Access Health Policy 2015, 3, 1. 10.3402/jmahp.v3.29414, https://pubmed.ncbi.nlm.nih.gov/27123186. - DOI - PMC - PubMed
-
- Hodgson J., Nat. Biotechnol. 2020, 38, 523. https://www.nature.com/articles/d41587-020-00005-z. - PubMed
-
- Amanat F., Krammer F., Immunity 2020, 52(4), 583. https://pubmed.ncbi.nlm.nih.gov/32259480. - PMC - PubMed
-
- Rauch S., Jasny E., Schmidt K. E., Petsch B., Front. Immunol. 2018, 9, 1963. https://www.ncbi.nlm.nih.gov/pubmed/30283434. - PMC - PubMed
LinkOut - more resources
Full Text Sources
