Implementing radical cure diagnostics for malaria: user perspectives on G6PD testing in Bangladesh
- PMID: 33980257
- PMCID: PMC8114691
- DOI: 10.1186/s12936-021-03743-w
Implementing radical cure diagnostics for malaria: user perspectives on G6PD testing in Bangladesh
Abstract
Background: The radical cure of Plasmodium vivax requires treatment with an 8-aminoquinoline drug, such as primaquine and tafenoquine, to eradicate liver hypnozoite stages, which can reactivate to cause relapsing infections. Safe treatment regimens require prior screening of patients for glucose-6-phosphate dehydrogenase (G6PD) deficiency to avoid potential life-threatening drug induced haemolysis. Testing is rarely available in malaria endemic countries, but will be needed to support routine use of radical cure. This study investigates end-user perspectives in Bangladesh on the introduction of a quantitative G6PD test (SD Biosensor STANDARD™ G6PD analyser) to support malaria elimination.
Methods: The perspectives of users on the SD Biosensor test were analysed using semi-structured interviews and focus group discussions with health care providers and malaria programme officers in Bangladesh. Key emerging themes regarding the feasibility of introducing this test into routine practice, including perceived barriers, were analysed.
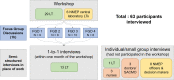
Results: In total 63 participants were interviewed. Participants emphasized the life-saving potential of the biosensor, but raised concerns including the impact of limited staff time, high workload and some technical aspects of the device. Participants highlighted that there are both too few and too many P. vivax patients to implement G6PD testing owing to challenges of funding, workload and complex testing infrastructure. Implementing the biosensor would require flexibility and improvisation to deal with remote sites, overcoming a low index of suspicion and mutual interplay of declining patient numbers and reluctance to test. This approach would generate new forms of evidence to justify introduction in policy and carefully consider questions of deployment given declining patient numbers.
Conclusions: The results of the study show that, in an elimination context, the importance of malaria needs to be maintained for both policy makers and the affected communities, in this case by ensuring P. vivax, PQ treatment, and G6PD deficiency remain visible. Availability of new technologies, such as the biosensor, will fuel ongoing debates about priorities for allocating resources that must be adapted to a constantly evolving target. Technical and logistical concerns regarding the biosensor should be addressed by future product designs, adequate training, strengthened supply chains, and careful planning of communication, advocacy and staff interactions at all health system levels.
Keywords: Bangladesh; G6PD testing; P. vivax; Point of care diagnostics; Users.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Barriers to routine G6PD testing prior to treatment with primaquine.Malar J. 2017 Aug 10;16(1):329. doi: 10.1186/s12936-017-1981-y. Malar J. 2017. PMID: 28797255 Free PMC article.
-
Challenges for achieving safe and effective radical cure of Plasmodium vivax: a round table discussion of the APMEN Vivax Working Group.Malar J. 2017 Apr 5;16(1):141. doi: 10.1186/s12936-017-1784-1. Malar J. 2017. PMID: 28381261 Free PMC article. Review.
-
Glucose 6 Phosphate Dehydrogenase (G6PD) quantitation using biosensors at the point of first contact: a mixed method study in Cambodia.Malar J. 2022 Oct 4;21(1):282. doi: 10.1186/s12936-022-04300-9. Malar J. 2022. PMID: 36195916 Free PMC article.
-
G6PD testing and radical cure for Plasmodium vivax in Cambodia: A mixed methods implementation study.PLoS One. 2022 Oct 20;17(10):e0275822. doi: 10.1371/journal.pone.0275822. eCollection 2022. PLoS One. 2022. PMID: 36264996 Free PMC article.
-
Contextual factors and G6PD diagnostic testing: a scoping review and evidence and gap map.Malar J. 2024 Aug 12;23(1):241. doi: 10.1186/s12936-024-05050-6. Malar J. 2024. PMID: 39135005 Free PMC article.
Cited by
-
Perspectives of healthcare professionals on training for quantitative G6PD testing during implementation of tafenoquine in Brazil (QualiTRuST Study).PLoS Negl Trop Dis. 2024 Jun 5;18(6):e0012197. doi: 10.1371/journal.pntd.0012197. eCollection 2024 Jun. PLoS Negl Trop Dis. 2024. PMID: 38837977 Free PMC article.
-
Clinical performance validation of the STANDARD G6PD test: A multi-country pooled analysis.PLoS Negl Trop Dis. 2023 Oct 12;17(10):e0011652. doi: 10.1371/journal.pntd.0011652. eCollection 2023 Oct. PLoS Negl Trop Dis. 2023. PMID: 37824592 Free PMC article.
-
Population screening for glucose-6-phosphate dehydrogenase deficiency using quantitative point-of-care tests: a systematic review.Front Genet. 2023 Jun 14;14:1098828. doi: 10.3389/fgene.2023.1098828. eCollection 2023. Front Genet. 2023. PMID: 37388931 Free PMC article.
-
Radical cure for Plasmodium vivax malaria after G6PD qualitative testing in four provinces in Cambodia, results from Phase I implementation.Malar J. 2024 Feb 23;23(1):56. doi: 10.1186/s12936-024-04884-4. Malar J. 2024. PMID: 38395925 Free PMC article.
-
Plasmodium vivax: the potential obstacles it presents to malaria elimination and eradication.Trop Dis Travel Med Vaccines. 2022 Dec 15;8(1):27. doi: 10.1186/s40794-022-00185-3. Trop Dis Travel Med Vaccines. 2022. PMID: 36522671 Free PMC article. Review.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

