Fas/FasL mediates NF-κBp65/PUMA-modulated hepatocytes apoptosis via autophagy to drive liver fibrosis
- PMID: 33980818
- PMCID: PMC8115181
- DOI: 10.1038/s41419-021-03749-x
Fas/FasL mediates NF-κBp65/PUMA-modulated hepatocytes apoptosis via autophagy to drive liver fibrosis
Abstract
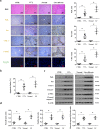
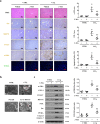
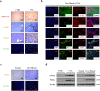
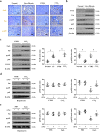
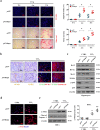
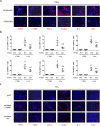
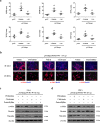
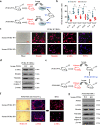
Fas/Fas ligand (FasL)-mediated cell apoptosis involves a variety of physiological and pathological processes including chronic hepatic diseases, and hepatocytes apoptosis contributes to the development of liver fibrosis following various causes. However, the mechanism of the Fas/FasL signaling and hepatocytes apoptosis in liver fibrogenesis remains unclear. The Fas/FasL signaling and hepatocytes apoptosis in liver samples from both human sections and mouse models were investigated. NF-κBp65 wild-type mice (p65f/f), hepatocytes specific NF-κBp65 deletion mice (p65Δhepa), p53-upregulated modulator of apoptosis (PUMA) wild-type (PUMA-WT) and PUMA knockout (PUMA-KO) littermate models, and primary hepatic stellate cells (HSCs) were also used. The mechanism underlying Fas/FasL-regulated hepatocytes apoptosis to drive HSCs activation in fibrosis was further analyzed. We found Fas/FasL promoted PUMA-mediated hepatocytes apoptosis via regulating autophagy signaling and NF-κBp65 phosphorylation, while inhibition of autophagy or PUMA deficiency attenuated Fas/FasL-modulated hepatocytes apoptosis and liver fibrosis. Furthermore, NF-κBp65 in hepatocytes repressed PUMA-mediated hepatocytes apoptosis via regulating the Bcl-2 family, while NF-κBp65 deficiency in hepatocytes promoted PUMA-mediated hepatocytes apoptosis and enhanced apoptosis-linked inflammatory response, which contributed to the activation of HSCs and liver fibrogenesis. These results suggest that Fas/FasL contributes to NF-κBp65/PUMA-modulated hepatocytes apoptosis via autophagy to enhance liver fibrogenesis, and this network could be a potential therapeutic target for liver fibrosis.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

