Sterol O-acyltransferase 2 chaperoned by apolipoprotein J facilitates hepatic lipid accumulation following viral and nutrient stresses
- PMID: 33980978
- PMCID: PMC8115332
- DOI: 10.1038/s42003-021-02093-2
Sterol O-acyltransferase 2 chaperoned by apolipoprotein J facilitates hepatic lipid accumulation following viral and nutrient stresses
Abstract
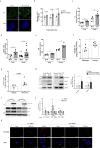
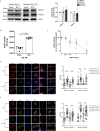
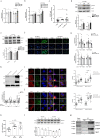
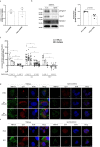
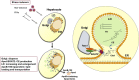
The risks of non-alcoholic fatty liver disease (NAFLD) include obese and non-obese stresses such as chronic hepatitis C virus (HCV) infection, but the regulatory determinants remain obscure. Apolipoprotein J (ApoJ) served as an ER-Golgi contact-site chaperone near lipid droplet (LD), facilitating HCV virion production. We hypothesized an interplay between hepatic ApoJ, cholesterol esterification and lipid deposit in response to NAFLD inducers. Exposures of HCV or free-fatty acids exhibited excess LDs along with increased ApoJ expression, whereas ApoJ silencing alleviated hepatic lipid accumulation. Both stresses could concomitantly disperse Golgi, induce closer ApoJ and sterol O-acyltransferase 2 (SOAT2) contacts via the N-terminal intrinsically disordered regions, and increase cholesteryl-ester. Furthermore, serum ApoJ correlated positively with cholesterol and low-density lipoprotein levels in normal glycaemic HCV patients, NAFLD patients and in mice with steatosis. Taken together, hepatic ApoJ might activate SOAT2 to supply cholesteryl-ester for lipid loads, thus providing a therapeutic target of stress-induced steatosis.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Apolipoprotein J, a glucose-upregulated molecular chaperone, stabilizes core and NS5A to promote infectious hepatitis C virus virion production.J Hepatol. 2014 Nov;61(5):984-93. doi: 10.1016/j.jhep.2014.06.026. Epub 2014 Jul 1. J Hepatol. 2014. PMID: 24996046
-
Sterol O-acyltransferase (SOAT/ACAT) activity is required to form cholesterol crystals in hepatocyte lipid droplets.Biochim Biophys Acta Mol Cell Biol Lipids. 2024 Aug;1869(6):159512. doi: 10.1016/j.bbalip.2024.159512. Epub 2024 May 16. Biochim Biophys Acta Mol Cell Biol Lipids. 2024. PMID: 38761895
-
Antagonizing apolipoprotein J chaperone promotes proteasomal degradation of mTOR and relieves hepatic lipid deposition.Hepatology. 2023 Oct 1;78(4):1182-1199. doi: 10.1097/HEP.0000000000000185. Epub 2023 Jan 3. Hepatology. 2023. PMID: 37013405
-
Pathophysiology of lipid droplet proteins in liver diseases.Exp Cell Res. 2016 Jan 15;340(2):187-92. doi: 10.1016/j.yexcr.2015.10.021. Epub 2015 Oct 26. Exp Cell Res. 2016. PMID: 26515554 Free PMC article. Review.
-
Hepatic lipid droplet homeostasis and fatty liver disease.Semin Cell Dev Biol. 2020 Dec;108:72-81. doi: 10.1016/j.semcdb.2020.04.011. Epub 2020 May 20. Semin Cell Dev Biol. 2020. PMID: 32444289 Review.
Cited by
-
Associations between serum clusterin levels and non-alcoholic fatty liver disease.Endocr Connect. 2023 Jun 12;12(7):e220545. doi: 10.1530/EC-22-0545. Print 2023 Jul 1. Endocr Connect. 2023. PMID: 37043769 Free PMC article.
-
Metabolic Dysfunction-Associated Steatotic Liver Disease: From a Very Low-Density Lipoprotein Perspective.Biomolecules. 2025 Jul 11;15(7):990. doi: 10.3390/biom15070990. Biomolecules. 2025. PMID: 40723862 Free PMC article. Review.
References
-
- Schade, M. et al. Cellular senescence and their role in liver metabolism in health and disease: overview and future directions. World J. Surg. Surgcal Res. 1, 1035 (2018).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

