Wheat phytase can alleviate the cellular toxic and inflammatory effects of lipopolysaccharide
- PMID: 33987589
- PMCID: PMC7882845
- DOI: 10.5187/jast.2021.e12
Wheat phytase can alleviate the cellular toxic and inflammatory effects of lipopolysaccharide
Abstract
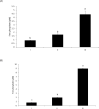
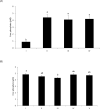
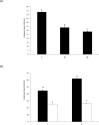
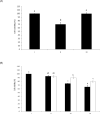
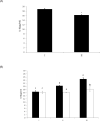
The objective of this study was to characterize the enzymatic hydrolysis of lipopolysaccharide (LPS) by wheat phytase and to investigate the effects of wheat phytase-treated LPS on in vitro toxicity, cell viability and release of a pro-inflammatory cytokine, interleukin (IL)-8 by target cells compared with the intact LPS. The phosphatase activity of wheat phytase towards LPS was investigated in the presence or absence of inhibitors such as L-phenylalanine and L-homoarginine. In vitro toxicity of LPS hydrolyzed with wheat phytase in comparison to intact LPS was assessed. Cell viability in human aortic endothelial (HAE) cells exposed to LPS treated with wheat phytase in comparison to intact LPS was measured. The release of IL-8 in human intestinal epithelial cell line, HT-29 cells applied to LPS treated with wheat phytase in comparison to intact LPS was assayed. Wheat phytase hydrolyzed LPS, resulting in a significant release of inorganic phosphate for 1 h (p < 0.05). Furthermore, the degradation of LPS by wheat phytase was nearly unaffected by the addition of L-phenylalanine, the inhibitor of tissue-specific alkaline phosphatase or L-homoarginine, the inhibitor of tissue-non-specific alkaline phosphatase. Wheat phytase effectively reduced the in vitro toxicity of LPS, resulting in a retention of 63% and 54% of its initial toxicity after 1-3 h of the enzyme reaction, respectively (p < 0.05). Intact LPS decreased the cell viability of HAE cells. However, LPS dephosphorylated by wheat phytase counteracted the inhibitory effect on cell viability. LPS treated with wheat phytase decreased IL-8 secretion from intestinal epithelial cell line, HT-29 cell to 14% (p < 0.05) when compared with intact LPS. In conclusion, wheat phytase is a potential therapeutic candidate and prophylactic agent for control of infections induced by pathogenic Gram-negative bacteria and associated LPS-mediated inflammatory diseases in animal husbandry.
Keywords: Inflammatory diseases; Lipopolysaccharide; Prophylactic agent; Therapeutic candidate; Wheat phytase.
© Copyright 2021 Korean Society of Animal Science and Technology.
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
Figures
References
-
- van Gucht S, van Reeth K, Pensaert M. Interaction between porcine reproductive-respiratory syndrome virus and bacterial endotoxin in the lungs of pigs: potentiation of cytokine production and respiratory disease. J Clin Microbiol. 2003;41:960–6. doi: 10.1128/JCM.41.3.960-966.2003. - DOI - PMC - PubMed
-
- Rauber RH, Perlin VJ, Fin CD, Mallmann AL, Miranda DP, Giacomini LZ, et al. Interference of Salmonella typhimurium lipopolysaccharide on performance and biological parameters of broiler chickens. Brazilian J Poultry Sci. 2014;16:77–81. doi: 10.1590/S1516-635X2014000100011. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

