Mitochondrial ATP fuels ABC transporter-mediated drug efflux in cancer chemoresistance
- PMID: 33990571
- PMCID: PMC8121950
- DOI: 10.1038/s41467-021-23071-6
Mitochondrial ATP fuels ABC transporter-mediated drug efflux in cancer chemoresistance
Abstract
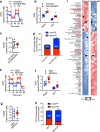
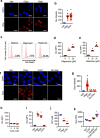
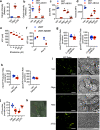
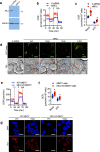
Chemotherapy remains the standard of care for most cancers worldwide, however development of chemoresistance due to the presence of the drug-effluxing ATP binding cassette (ABC) transporters remains a significant problem. The development of safe and effective means to overcome chemoresistance is critical for achieving durable remissions in many cancer patients. We have investigated the energetic demands of ABC transporters in the context of the metabolic adaptations of chemoresistant cancer cells. Here we show that ABC transporters use mitochondrial-derived ATP as a source of energy to efflux drugs out of cancer cells. We further demonstrate that the loss of methylation-controlled J protein (MCJ) (also named DnaJC15), an endogenous negative regulator of mitochondrial respiration, in chemoresistant cancer cells boosts their ability to produce ATP from mitochondria and fuel ABC transporters. We have developed MCJ mimetics that can attenuate mitochondrial respiration and safely overcome chemoresistance in vitro and in vivo. Administration of MCJ mimetics in combination with standard chemotherapeutic drugs could therefore become an alternative strategy for treatment of multiple cancers.
Conflict of interest statement
M. Rincon and T. Thornton have a patent application related to this work. K.F. was supported by Mitotherapeutics LLC. M. Rincon is a co-founder and a member of the Scientific Advisory Board of Mitotherapeutix LLC. The other authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

