Technological tools and strategies for culturing human gut microbiota in engineered in vitro models
- PMID: 33990954
- PMCID: PMC8361989
- DOI: 10.1002/bit.27816
Technological tools and strategies for culturing human gut microbiota in engineered in vitro models
Abstract
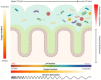
The gut microbiota directly impacts the pathophysiology of different human body districts. Consequently, microbiota investigation is an hot topic of research and its in vitro culture has gained extreme interest in different fields. However, the high sensitivity of microbiota to external stimuli, such as sampling procedure, and the physicochemical complexity of the gut environment make its in vitro culture a challenging task. New engineered microfluidic gut-on-a-chip devices have the potential to model some important features of the intestinal structure, but they are usually unable to sustain culture of microbiota over an extended period of time. The integration of gut-on-a-chip devices with bioreactors for continuous bacterial culture would lead to fast advances in the study of microbiota-host crosstalk. In this review, we summarize the main technologies for the continuous culture of microbiota as upstream systems to be coupled with microfluidic devices to study bacteria-host cells communication. The engineering of integrated microfluidic platforms, capable of sustaining both anaerobic and aerobic cultures, would be the starting point to unveil complex biological phenomena proper of the microbiota-host crosstalks, paving to way to multiple research and technological applications.
Keywords: anaerobiosis; bioreactors; gut-brain axis; microfluidic systems; organ-on-a-chip.
© 2021 The Authors. Biotechnology and Bioengineering Published by Wiley Periodicals LLC.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

