New methylene blue derivatives suggest novel anti-orthopoxviral strategies
- PMID: 33992710
- PMCID: PMC9629033
- DOI: 10.1016/j.antiviral.2021.105086
New methylene blue derivatives suggest novel anti-orthopoxviral strategies
Abstract
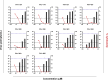
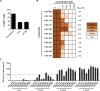
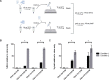
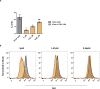
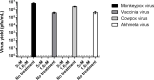
Decades after the eradication of smallpox and the discontinuation of routine smallpox vaccination, over half of the world's population is immunologically naïve to variola virus and other orthopoxviruses (OPXVs). Even in those previously vaccinated against smallpox, protective immunity wanes over time. As such, there is a concomitant increase in the incidence of human OPXV infections worldwide. To identify novel antiviral compounds with potent anti-OPXV potential, we characterized the inhibitory activity of PAV-866 and other methylene blue derivatives against the prototypic poxvirus, vaccinia virus (VACV). These compounds inactivated virions prior to infection and consequently inhibited viral binding, fusion and entry. The compounds exhibited strong virucidal activity at non-cytotoxic concentrations, and inhibited VACV infection when added before, during or after viral adsorption. The compounds were effective against other OPXVs including monkeypox virus, cowpox virus and the newly identified Akhmeta virus. Altogether, these findings reveal a novel mode of inhibition that has not previously been demonstrated for small molecule compounds against VACV. Additional studies are in progress to determine the in vivo efficacy of these compounds against OPXVs and further characterize the anti-viral effects of these derivatives.
Keywords: Antiviral; Methylene blue; Novel inhibitor; PAV-866; Poxviruses; Vaccinia virus.
Copyright © 2021 The Author(s). Published by Elsevier B.V. All rights reserved.
Figures
References
-
- Bisbal C., Martinand C., Silhol M., Lebleu B., Salehzada T. Cloning and characterization of a RNAse L inhibitor. A new component of the interferon-regulated 2-5A pathway. J. Biol. Chem. 1995;270:13308–13317. - PubMed
-
- Chan-Tack K.M., Harrington P.R., Choi S.Y., Myers L., O'Rear J., Seo S., McMillan D., Ghantous H., Birnkrant D., Sherwat A.I. Assessing a drug for an eradicated human disease: US Food and Drug Administration review of tecovirimat for the treatment of smallpox. Lancet Infect. Dis. 2019;19:e221–e224. - PubMed
-
- Cryer M., Lane K., Greer M., Cates R., Burt S., Andrus M., Zou J., Rogers P., Hansen M.D., Burgado J., Panayampalli S.S., Day C.W., Smee D.F., Johnson B.F. Isolation and identification of compounds from Kalanchoe pinnata having human alphaherpesvirus and vaccinia virus antiviral activity. Pharm. Biol. 2017;55:1586–1591. - PMC - PubMed
-
- Damon I. In: sixth ed. Knipe D.M., Howley P.M., editors. vol. 2. Lippincott, Williams and Wilkins; Philadelphia, pA, USA: 2014. Poxviruses; p. 2160. (Fields Virology).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

