Lipid-nanoparticle-encapsulated mRNA vaccines induce protective memory CD8 T cells against a lethal viral infection
- PMID: 33992803
- PMCID: PMC8417516
- DOI: 10.1016/j.ymthe.2021.05.011
Lipid-nanoparticle-encapsulated mRNA vaccines induce protective memory CD8 T cells against a lethal viral infection
Abstract
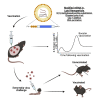
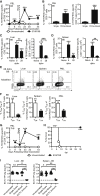
It is well established that memory CD8 T cells protect susceptible strains of mice from mousepox, a lethal viral disease caused by ectromelia virus (ECTV), the murine counterpart to human variola virus. While mRNA vaccines induce protective antibody (Ab) responses, it is unknown whether they also induce protective memory CD8 T cells. We now show that immunization with different doses of unmodified or N(1)-methylpseudouridine-modified mRNA (modified mRNA) in lipid nanoparticles (LNP) encoding the ECTV gene EVM158 induced similarly strong CD8 T cell responses to the epitope TSYKFESV, albeit unmodified mRNA-LNP had adverse effects at the inoculation site. A single immunization with 10 μg modified mRNA-LNP protected most susceptible mice from mousepox, and booster vaccination increased the memory CD8 T cell pool, providing full protection. Moreover, modified mRNA-LNP encoding TSYKFESV appended to green fluorescent protein (GFP) protected against wild-type ECTV infection while lymphocytic choriomeningitis virus glycoprotein (GP) modified mRNA-LNP protected against ECTV expressing GP epitopes. Thus, modified mRNA-LNP can be used to create protective CD8 T cell-based vaccines against viral infections.
Keywords: CD8 T cells; lipid nanoparticle; mRNA vaccine; modified mRNA; poxvirus; virus.
Copyright © 2021 The American Society of Gene and Cell Therapy. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
N.P. and D.W. are named on patents that describe the use of nucleoside-modified mRNA as a platform to deliver therapeutic proteins and vaccines. They have disclosed those interests fully to the University of Pennsylvania and have in place an approved plan for managing any potential conflicts arising from licensing of the patents. P.J.C.L. and Y.K.T. are employees of Acuitas Therapeutics, a company focused on the development of lipid nanoparticulate nucleic acid delivery systems for therapeutic applications and are named on patent applications describing lipid nanoparticles for delivery of mRNA.
Figures




Similar articles
-
The roles of CD4+ T cell help, sex, and dose in the induction of protective CD8+ T cells against a lethal poxvirus by mRNA-LNP vaccines.Mol Ther Nucleic Acids. 2024 Jul 20;35(3):102279. doi: 10.1016/j.omtn.2024.102279. eCollection 2024 Sep 10. Mol Ther Nucleic Acids. 2024. PMID: 39188304 Free PMC article.
-
Loss of Resistance to Mousepox during Chronic Lymphocytic Choriomeningitis Virus Infection Is Associated with Impaired T-Cell Responses and Can Be Rescued by Immunization.J Virol. 2020 Feb 14;94(5):e01832-19. doi: 10.1128/JVI.01832-19. Print 2020 Feb 14. J Virol. 2020. PMID: 31826990 Free PMC article.
-
Memory CD8+ T cells specific for a single immunodominant or subdominant determinant induced by peptide-dendritic cell immunization protect from an acute lethal viral disease.J Virol. 2012 Sep;86(18):9748-59. doi: 10.1128/JVI.00981-12. Epub 2012 Jun 27. J Virol. 2012. PMID: 22740418 Free PMC article.
-
The Pathogenesis and Immunobiology of Mousepox.Adv Immunol. 2016;129:251-76. doi: 10.1016/bs.ai.2015.10.001. Epub 2015 Nov 21. Adv Immunol. 2016. PMID: 26791861 Review.
-
Ectromelia virus: the causative agent of mousepox.J Gen Virol. 2005 Oct;86(Pt 10):2645-2659. doi: 10.1099/vir.0.81090-0. J Gen Virol. 2005. PMID: 16186218 Review.
Cited by
-
The roles of CD4+ T cell help, sex, and dose in the induction of protective CD8+ T cells against a lethal poxvirus by mRNA-LNP vaccines.Mol Ther Nucleic Acids. 2024 Jul 20;35(3):102279. doi: 10.1016/j.omtn.2024.102279. eCollection 2024 Sep 10. Mol Ther Nucleic Acids. 2024. PMID: 39188304 Free PMC article.
-
A highly stable lyophilized mRNA vaccine for Herpes Zoster provides potent cellular and humoral responses.NPJ Vaccines. 2025 Mar 14;10(1):49. doi: 10.1038/s41541-025-01093-1. NPJ Vaccines. 2025. PMID: 40087280 Free PMC article.
-
Enhancing Skin Cancer Immunotheranostics and Precision Medicine through Functionalized Nanomodulators and Nanosensors: Recent Development and Prospects.Int J Mol Sci. 2023 Feb 9;24(4):3493. doi: 10.3390/ijms24043493. Int J Mol Sci. 2023. PMID: 36834917 Free PMC article. Review.
-
Circular RNA vaccine in disease prevention and treatment.Signal Transduct Target Ther. 2023 Sep 11;8(1):341. doi: 10.1038/s41392-023-01561-x. Signal Transduct Target Ther. 2023. PMID: 37691066 Free PMC article. Review.
-
Carrier-free mRNA vaccine induces robust immunity against SARS-CoV-2 in mice and non-human primates without systemic reactogenicity.Mol Ther. 2024 May 1;32(5):1266-1283. doi: 10.1016/j.ymthe.2024.03.022. Epub 2024 Apr 2. Mol Ther. 2024. PMID: 38569556 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

