β-Cell failure in diabetes: Common susceptibility and mechanisms shared between type 1 and type 2 diabetes
- PMID: 33993642
- PMCID: PMC8409822
- DOI: 10.1111/jdi.13576
β-Cell failure in diabetes: Common susceptibility and mechanisms shared between type 1 and type 2 diabetes
Abstract
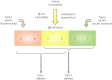
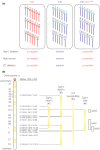
Diabetes mellitus is etiologically classified into type 1, type 2 and other types of diabetes. Despite distinct etiologies and pathogenesis of these subtypes, many studies have suggested the presence of shared susceptibilities and underlying mechanisms in β-cell failure among different types of diabetes. Understanding these susceptibilities and mechanisms can help in the development of therapeutic strategies regardless of the diabetes subtype. In this review, we discuss recent evidence indicating the shared genetic susceptibilities and common molecular mechanisms between type 1, type 2 and other types of diabetes, and highlight the future prospects as well.
Keywords: Endoplasmic reticulum stress; Oxidative stress; β-Cell failure.
© 2021 The Authors. Journal of Diabetes Investigation published by Asian Association for the Study of Diabetes (AASD) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
[Chronicle of a death foretold: endoplasmic reticulum stress and beta cell apoptosis in diabetes].Med Sci (Paris). 2014 May;30(5):496-9. doi: 10.1051/medsci/20143005007. Epub 2014 Jun 13. Med Sci (Paris). 2014. PMID: 24939531 French. No abstract available.
-
Pancreatic Beta Cell Death: Novel Potential Mechanisms in Diabetes Therapy.J Diabetes Res. 2018 Feb 19;2018:9601801. doi: 10.1155/2018/9601801. eCollection 2018. J Diabetes Res. 2018. PMID: 29670917 Free PMC article. Review.
-
Stress-Induced Translational Regulation Mediated by RNA Binding Proteins: Key Links to β-Cell Failure in Diabetes.Diabetes. 2020 Apr;69(4):499-507. doi: 10.2337/dbi18-0068. Diabetes. 2020. PMID: 32198193 Free PMC article. Review.
-
Oxidative and endoplasmic reticulum stress in β-cell dysfunction in diabetes.J Mol Endocrinol. 2016 Feb;56(2):R33-54. doi: 10.1530/JME-15-0232. Epub 2015 Nov 17. J Mol Endocrinol. 2016. PMID: 26576641 Review.
-
β Cell Senescence as a Common Contributor to Type 1 and Type 2 Diabetes.Trends Mol Med. 2019 Sep;25(9):735-737. doi: 10.1016/j.molmed.2019.07.009. Epub 2019 Aug 15. Trends Mol Med. 2019. PMID: 31422036
Cited by
-
Insight into continuous glucose monitoring: from medical basics to commercialized devices.Mikrochim Acta. 2023 Apr 6;190(5):177. doi: 10.1007/s00604-023-05743-w. Mikrochim Acta. 2023. PMID: 37022500 Review.
-
Effect of SPTLC1 on type 2 diabetes mellitus.World J Diabetes. 2025 Feb 15;16(2):94861. doi: 10.4239/wjd.v16.i2.94861. World J Diabetes. 2025. PMID: 39959268 Free PMC article.
-
Rapid and Slow Progressors Toward β-Cell Depletion and Their Predictors in Type 1 Diabetes: Prospective Longitudinal Study in Japanese Type 1 Diabetes (TIDE-J).Diabetes Care. 2025 Aug 1;48(8):1438-1445. doi: 10.2337/dc25-0579. Diabetes Care. 2025. PMID: 40509916 Free PMC article.
-
Antidiabetic Properties of Chitosan and Its Derivatives.Mar Drugs. 2022 Dec 17;20(12):784. doi: 10.3390/md20120784. Mar Drugs. 2022. PMID: 36547931 Free PMC article. Review.
-
Insulin-dependent diabetes mellitus in older adults: Current status and future prospects.Geriatr Gerontol Int. 2022 Aug;22(8):549-553. doi: 10.1111/ggi.14414. Epub 2022 Jun 16. Geriatr Gerontol Int. 2022. PMID: 35711119 Free PMC article. Review.
References
-
- American Diabetes Association . Diagnosis and classification of diabetes mellitus. Diabetes Care 2014; 37(suppl 1): S81–S90. - PubMed
-
- Ikegami H, Fujisawa T, Ogihara T. Mouse models of type 1 and type 2 diabetes derived from the same closed colony: genetic susceptibility shared between two types of diabetes. ILAR J 2004; 45: 268–277. - PubMed
-
- Boitard C, Efendic S, Ferrannini E, et al. A tale of two cousins: type 1 and type 2 diabetes. Diabetes 2005; 54(Suppl2): S1–S3. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

