Chronic Ethanol Exposures Leads to a Negative Affective State in Female Rats That Is Accompanied by a Paradoxical Decrease in Ventral Hippocampus Excitability
- PMID: 33994940
- PMCID: PMC8119765
- DOI: 10.3389/fnins.2021.669075
Chronic Ethanol Exposures Leads to a Negative Affective State in Female Rats That Is Accompanied by a Paradoxical Decrease in Ventral Hippocampus Excitability
Abstract
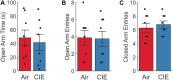
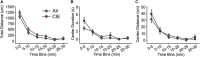
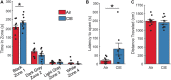
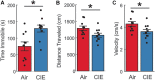
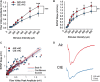
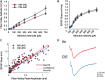
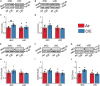
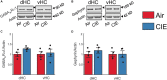
Alcohol use disorder (AUD) differentially impacts men and women and a growing body of evidence points to sex-dependent adaptations in a number of brain regions. In a prior study, we explored the effect of a chronic intermittent ethanol exposure (CIE) model of AUD on neuronal and molecular adaptations in the dorsal and ventral domains of the hippocampus (dHC and vHC, respectively) in male rats. We found the vHC to be particularly sensitive to CIE, showing an increase in neuronal excitability and synaptic proteins associated with augmented excitation. These findings were accompanied by a CIE-dependent increase in anxiety-like behaviors. To explore sex-dependent adaptations in the hippocampus, we conducted a similar study in female rats. CIE-treated female rats showed a relatively modest increase in anxiety-like behaviors along with a robust increase in depressive-like measures. Despite both sexes showing clear evidence of a negative affective state following CIE, the vHC of females showed a decrease, rather than an increase, in neuronal excitability. In line with the reduced sensitivity to neural adaptations in the dHC of male rats, we were unable to identify any functional changes in the dHC of females. The functional changes of the vHC in female rats could not be explained by altered expression levels of a number of proteins typically associated with changes in neuronal excitability. Taken together, these findings point to sex as a major factor in CIE-dependent hippocampal adaptations that should be explored further to better understand possible gender differences in the etiology and treatment of AUD.
Keywords: alcohol use disorder; chronic intermittent ethanol exposure; electrophysiology; hippocampus; negative affective behavior.
Copyright © 2021 Bach, Morgan, Ewin, Barth, Raab-Graham and Weiner.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Annamneedi A., Caliskan G., Muller S., Montag D., Budinger E., Angenstein F., et al. (2018). Ablation of the presynaptic organizer Bassoon in excitatory neurons retards dentate gyrus maturation and enhances learning performance. Brain Struct. Funct. 223 3423–3445. 10.1007/s00429-018-1692-3 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

