An Evolutionary Microcircuit Approach to the Neural Basis of High Dimensional Sensory Processing in Olfaction
- PMID: 33994949
- PMCID: PMC8120314
- DOI: 10.3389/fncel.2021.658480
An Evolutionary Microcircuit Approach to the Neural Basis of High Dimensional Sensory Processing in Olfaction
Abstract
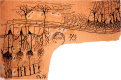
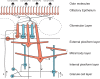
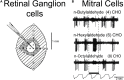
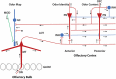
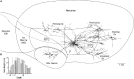
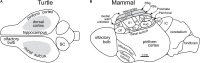
Odor stimuli consist of thousands of possible molecules, each molecule with many different properties, each property a dimension of the stimulus. Processing these high dimensional stimuli would appear to require many stages in the brain to reach odor perception, yet, in mammals, after the sensory receptors this is accomplished through only two regions, the olfactory bulb and olfactory cortex. We take a first step toward a fundamental understanding by identifying the sequence of local operations carried out by microcircuits in the pathway. Parallel research provided strong evidence that processed odor information is spatial representations of odor molecules that constitute odor images in the olfactory bulb and odor objects in olfactory cortex. Paleontology provides a unique advantage with evolutionary insights providing evidence that the basic architecture of the olfactory pathway almost from the start ∼330 million years ago (mya) has included an overwhelming input from olfactory sensory neurons combined with a large olfactory bulb and olfactory cortex to process that input, driven by olfactory receptor gene duplications. We identify a sequence of over 20 microcircuits that are involved, and expand on results of research on several microcircuits that give the best insights thus far into the nature of the high dimensional processing.
Keywords: content addressable memory; evolution; lateral inhibition; mammalian paleontology; microcircuit; neuronal microcircuits; olfactory processing; sensory dimensionality.
Copyright © 2021 Shepherd, Rowe and Greer.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Allison A. C. (1953). The morphology of the olfactory system in vertebrates. Biol. Rev. 28 195–277. 10.1111/j.1469-185x.1953.tb01376.x - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

