Microbial Degradation of Citric Acid in Low Level Radioactive Waste Disposal: Impact on Biomineralization Reactions
- PMID: 33995289
- PMCID: PMC8114274
- DOI: 10.3389/fmicb.2021.565855
Microbial Degradation of Citric Acid in Low Level Radioactive Waste Disposal: Impact on Biomineralization Reactions
Abstract
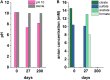
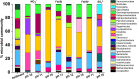
Organic complexants are present in some radioactive wastes and can challenge waste disposal as they may enhance subsurface mobility of radionuclides and contaminant species via chelation. The principal sources of organic complexing agents in low level radioactive wastes (LLW) originate from chemical decontamination activities. Polycarboxylic organic decontaminants such as citric and oxalic acid are of interest as currently there is a paucity of data on their biodegradation at high pH and under disposal conditions. This work explores the biogeochemical fate of citric acid, a model decontaminant, under high pH anaerobic conditions relevant to disposal of LLW in cementitious disposal environments. Anaerobic microcosm experiments were set up, using a high pH adapted microbial inoculum from a well characterized environmental site, to explore biodegradation of citrate under representative repository conditions. Experiments were initiated at three different pH values (10, 11, and 12) and citrate was supplied as the electron donor and carbon source, under fermentative, nitrate-, Fe(III)- and sulfate- reducing conditions. Results showed that citrate was oxidized using nitrate or Fe(III) as the electron acceptor at > pH 11. Citrate was fully degraded and removed from solution in the nitrate reducing system at pH 10 and pH 11. Here, the microcosm pH decreased as protons were generated during citrate oxidation. In the Fe(III)-reducing systems, the citrate removal rate was slower than in the nitrate reducing systems. This was presumably as Fe(III)-reduction consumes fewer moles of citrate than nitrate reduction for the same molar concentrations of electron acceptor. The pH did not change significantly in the Fe(III)-reducing systems. Sulfate reduction only occurred in a single microcosm at pH 10. Here, citrate was fully removed from solution, alongside ingrowth of acetate and formate, likely fermentation products. The acetate and lactate were subsequently used as electron donors during sulfate-reduction and there was an associated decrease in solution pH. Interestingly, in the Fe(III) reducing experiments, Fe(II) ingrowth was observed at pH values recorded up to 11.7. Here, TEM analysis of the resultant solid Fe-phase indicated that nanocrystalline magnetite formed as an end product of Fe(III)-reduction under these extreme conditions. PCR-based high-throughput 16S rRNA gene sequencing revealed that bacteria capable of nitrate Fe(III) and sulfate reduction became enriched in the relevant, biologically active systems. In addition, some fermentative organisms were identified in the Fe(III)- and sulfate-reducing systems. The microbial communities present were consistent with expectations based on the geochemical data. These results are important to improve long-term environmental safety case development for cementitious LLW waste disposal.
Keywords: alkaline; anaerobic biodegradation; biodegradation; bioreduction; citric acid; complexing agent; high pH; low level radioactive waste.
Copyright © 2021 Byrd, Lloyd, Small, Taylor, Bagshaw, Boothman and Morris.
Conflict of interest statement
The authors declare that this study received funding from Low Level Waste Repository Ltd. The funder had the following involvement with the study: support and contextualization for experimental design, manuscript review. The funder was not involved in the collection, analysis and interpretation of data, or, the decision to submit it for publication. FT was employed by company Low Level Waste Repository Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Akers D. W., Kraft N. C., Mandler J. W. (1994a). Compression and Immersion Tests and Leaching of Radionuclides, Stable Metals, and Chelating Agents from Cement-Solidified Decontamination Waste Collected from Nuclear Power Stations. NUREG/CR-6201, EGG-2736. Washington, D.C: Nuclear Regulatory Commission.
-
- Akers D. W., Kraft N. C., Mandler J. W. (1994b). Release of Radionuclides and Chelating Agents from Cement Solidified Decontamination Low-Level Radioactive Waste Collected from the Peach Bottom Atomic Power Station Unit 3. NUREG/CR-6164, EGG-2722. Washington, D.C: Nuclear Regulatory Commission.
-
- Albrecht A., Bertron A., Libert M. (2013). “Microbial catalysis of redox reactions in concrete cells of nuclear waste repositories: a review and introduction,” in Cement-Based Materials for Nuclear Waste Storage, eds Bart F., Cau-di-Coumes C., Frizon F. (New York, NY: Springer; ), 147–159. 10.1007/978-1-4614-3445-0 - DOI
-
- Ashbrook A. W. (1986). The refining and conversion of uranium yellowcake to uranium dioxide and uranium hexafluoride fuels in Canada: current processes. Iaea Tecdoc. 420:9.
LinkOut - more resources
Full Text Sources
Other Literature Sources

